A Guest Post by SK
SK is a professor emeritus in the department of Mechanical and Aeronautical Engineering at a Major University in the USA.
Corrections to the first three equations were made on Feb 25, 2017.
The report reviewed here claims to rely on thermodynamics arguments to predict oil’s price-volume trajectory going forward.
Classical Thermodynamic analysis
First a few lines about thermodynamic analysis. The early contributions to thermodynamics by Carnot, a military engineer by training, were based on study of heat engines. The same theme was followed by Clausius, Kelvin, Planck and others. The study of heat engines is still an important aspect of mechanical engineering and as such appears in engineering thermodynamic textbooks with one chapter devoted to the analysis steam power plants, and another on the thermodynamic cycles that model, spark ignition engines, diesel engines, and gas turbine power plants. Similar analysis is next extended to refrigeration cycles and performance of heat pumps.
The fundamental analysis is based on the first law of thermodynamics together with a mass balance. The second law of thermodynamics introduces the entropy as a thermodynamic property and the related concepts of reversible processes and reversible heat transfer. Irreversibilities of real processes are taken into account by assigning a value of experimentally determined efficiency to equipment such as pumps, compressors and turbines and this way the reversible processes are related to the actual ones.
A relatively recent development has been a systematic use of an exergy balance to examine where in a complex energy system irreversibilities take place. Exergy is defined as the maximum theoretical work that can be obtained from a system and its environment as the system comes to equilibrium with its environment. By combining the first and second laws of thermodynamics an exergy balance can be written down. Rudimentary exergy analysis can be found in the 1941 book Thermodynamics by Joseph Keenan. It was called availability analysis at that time. The most systematic development of the exergy analysis is in the textbook Fundamentals of Engineering Thermodynamics by M. Moran, H. Shapiro, D. Boettner and M. Bailey, 7th ed. John Wiley, 2011.
Developments over the last century have led to increases in the thermodynamic efficiency of systems such as a coal or nuclear power plants, mainly by increasing the maximum steam temperature of the plant. As the hot steam from a boiler or a superheater flows into the steam turbine, the first row of blades encounters a hot environment. This requires that these blades be made of materials that withstand the stresses generated at these temperatures. Such developments have increased the maximum temperature of these power plants to about 1000 F, but further improvements have stalled over the last half a century. To be sure, in the interim feedwater heaters and reheating have been used to increase the efficiency of the plant. For gas fired power plants combustion temperature is higher and turbine designers implement both cooling technology and use high temperature materials for the blades. Today they are made of single crystals, capable of withstanding the hot combustion gases.
Although the entropy balance equation can be used (although typically only for steady state systems) to determine the entropy production, to carry it out requires that sufficient number of thermodynamic properties and interactions are known at the system boundaries. Since such a calculation needs to be carried out after the thermodynamic analysis has been completed, it is seldom done in engineering practice because the knowledge of the same properties allows also the determination of the thermodynamic efficiency of the system.
The advocates of exergy accounting claim that knowing where the exergy destruction takes place in a system is a good way of allocating development money to improve it. This kind of analysis has not taken hold in industry either, simply because, manufacturers of, say turbines, know that the efficiency of the turbine is a measure of the irreversibilities and they direct their efforts toward understanding how the blades of the turbine can be shaped in order to reduce these irreversibilities. Such a task is based on aerodynamic calculations. Compressors and pump are, by the nature of the flow through them, machines with lower efficiency than turbines and their improvement requires again experts with fluid dynamic knowledge to improve them. Similarly improving the heat transfer in a heat exchanger is carried out by making improvements in the heat exchanger surfaces and reducing pressure losses. If these improve the heat transfer, the entropy production is reduced. Here the expertise of a heat transfer specialist rather than a thermodynamicists is needed.
One interesting application of exergy analysis is to calculate the second law efficiency. A high second law efficiency means that the source of energy is well matched with the application. Thus heating shower water with a thermal solar heater is a good match, as unfocused solar energy raises the water temperature high enough to serve as shower water, but not nearly so high as to create superheated steam to power a steam turbine. Thus the most important insight to be obtained is to match the source of energy to the application, and once this insight is internalized, calculation of the second law efficiency adds only marginally to understanding. For this reason it is seldom used in industry. To be sure, optimization of a system’s second law efficiency is still worthwhile, but using other metrics this can be done with topics based on heat transfer and fluid dynamics, with stress analysis, material selection and related fields as further aids. Interestingly exergy analysis shows that most of the exergy destruction takes place in the combustion of the fuel, but there is not much one can do to reduce this destruction. For this reason a naive application of exergy analysis may lead the poor allocation of development funds. Besides, the manufacturer of the turbine does not design heat exchangers so their coordination would be difficult to carry out.
Thermodynamic analysis in the report by the Hill’s Group
The report uses the second law of thermodynamics as the starting point. The unsteady entropy balance for a control volume with one exit and no inlet is given as
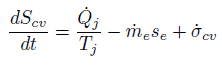
Next comes the assumption that at all times
![]() It is based on the observation that because at the end of oil production when the reservoir has been completely depleted the flow will stop and nothing much takes place, then both of these terms are zero. After cancelling these terms the entropy production is seen to be related to the heat transfer. But his assumption is clearly unjustified while the oil is being extracted and these two terms do not cancel each other. The neglect of the terms leads to an equation that omits the entropy production that is caused by the irreversibilities of the oil flow through the permeable reservoir rock.
It is based on the observation that because at the end of oil production when the reservoir has been completely depleted the flow will stop and nothing much takes place, then both of these terms are zero. After cancelling these terms the entropy production is seen to be related to the heat transfer. But his assumption is clearly unjustified while the oil is being extracted and these two terms do not cancel each other. The neglect of the terms leads to an equation that omits the entropy production that is caused by the irreversibilities of the oil flow through the permeable reservoir rock.
The incorrect canceling leads to the equation
![]() and the former choice assumes that the entropy production is known and then this equation is used to calculate the heat transfer. If on the other hand, the aim is to calculate the entropy production in the reservoir, there is no indication in the report how the heat transfer is calculated. In thinking about the heat transfer, for a control volume that includes the reservoir only, it appears that the heat interaction between the system and the surroundings is mainly caused by the geothermal gradient. That is, heat enters from the lower boundary and leaves across the upper boundary. This is a passive process. The fact that the oil and water in the reservoir have some average temperature in the geological setting only influences the viscosity of the fluids and thus how well they move through the reservoir, but from the energetic standpoint the sensible energy is not important. That is, there is no attempt made to extract this energy in a heat exchanger, nor is the high pressure used to extract energy in an expander. Rather the oil and water mixture flows through a set of throttling valves, in which the exergy is destroyed.
and the former choice assumes that the entropy production is known and then this equation is used to calculate the heat transfer. If on the other hand, the aim is to calculate the entropy production in the reservoir, there is no indication in the report how the heat transfer is calculated. In thinking about the heat transfer, for a control volume that includes the reservoir only, it appears that the heat interaction between the system and the surroundings is mainly caused by the geothermal gradient. That is, heat enters from the lower boundary and leaves across the upper boundary. This is a passive process. The fact that the oil and water in the reservoir have some average temperature in the geological setting only influences the viscosity of the fluids and thus how well they move through the reservoir, but from the energetic standpoint the sensible energy is not important. That is, there is no attempt made to extract this energy in a heat exchanger, nor is the high pressure used to extract energy in an expander. Rather the oil and water mixture flows through a set of throttling valves, in which the exergy is destroyed.
If on the other hand the entropy production were known independently, then this equation could be used to calculate the heat transfer, but the answer would be incorrect because entropy production is caused by both heat transfer and irreversible processes taking place inside the control volume. For the control volume consisting of the reservoir, entropy production takes place mainly in the pores of the permeable reservoir rock as the flow is forced out. This takes place by viscous dissipation and although it can be calculated in principle, in practice such a calculation is nearly impossible to carry out from first principles. The entropy production rate for the system would then be calculated by integration of the local values over the entire reservoir.
There does not seem to be much point in attempting to relate the entropy of crude oil in the reservoir to that of the output stream. Whereas entropy of pure substances are tabulated in thermodynamic tables, for mixtures such as crude oil it is not known. It appears that the attempt in the report is to relate the efficiency of oil extraction to the reversible work needed to accomplish the same task.
Next in the analysis is a calculation of ETp. It is defined as the total production energy, or the total work required to extract, process, and distribute a volumetric quantity (a gallon) of crude oil. The report offers the equation
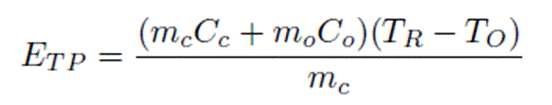
as a way to calculate it. But this is the energy of the sensible part of the oil-water mixture above the reference temperature To. It does not include the chemical energy of the crude oil and the formula cannot be reconciled with the definition of ETp.
The following equation also appears in the report
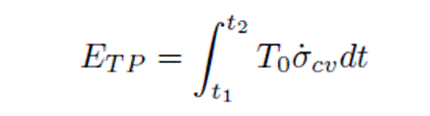 Thus there are two equations to use for calculating ETp and there is no mention what the independent variables are and what is calculated using these equations. If the value of ETp is calculated this way then how is the previous equation used? The only unknowns are the reservoir temperature Tr and the oil-water ratio, if the total flow rate is determined from the depletion rate equation. The reservoir temperature can be measured, so the unknown seems to be the water oil ratio. However, the report makes use of an empirical equation for the oil/water ratio as a function of the percent depletion of the reservoir. Finally the last equation can only be used to calculate the change in exergy, and this would necessitate a new symbol to be introduced for exergy, e.g. XTp, for exergy is not the same as energy.
Thus there are two equations to use for calculating ETp and there is no mention what the independent variables are and what is calculated using these equations. If the value of ETp is calculated this way then how is the previous equation used? The only unknowns are the reservoir temperature Tr and the oil-water ratio, if the total flow rate is determined from the depletion rate equation. The reservoir temperature can be measured, so the unknown seems to be the water oil ratio. However, the report makes use of an empirical equation for the oil/water ratio as a function of the percent depletion of the reservoir. Finally the last equation can only be used to calculate the change in exergy, and this would necessitate a new symbol to be introduced for exergy, e.g. XTp, for exergy is not the same as energy.
The report next presents calculation of the oil extraction trajectory that is based on Hubbert’s methodology. The calculations are in close agreement what others have found, with cumulative production 2357 Gb that is somewhat larger than Campbell and Laherrere’s value of 2123 Gb. It is now well known that in the calculations based on logistic equation, there is a slow drift to large values of the ultimate production as more data has been included in the calculations with the passing of the years.
In the same section is also a discussion of the surface water cut as a function of the percent of oil extracted from a reservoir. The curve is then rotated in order to satisfy two criteria set by the authors. Now a rotation of a curve is a mathematical transformation and a curve cannot be arbitrarily rotated without destroying the underlying mathematical theory. Furthermore, the report states that ETp cannot exceed EG, the crude oil’s specific exergy. With the calculation of ETp suspect, this condition is also meaningless. Here again energy and exergy are used interchangeably.
Returning to the calculation in Section 4.1 of the report for calculating ETp by the equation
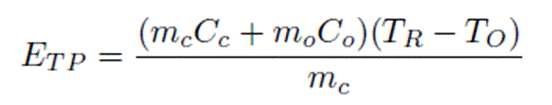
The statement on top of page 19 suggests that the water cut is an input parameter, in which case the value of ETp depends only on the reservoir temperature. The reservoir temperature in turn is a function of the depth of the well, owing to the geothermal gradient. This would allow this equation to be used to calculate the sensible energy of oil-water mixture. But what purpose does this serve? The sensible energy of the crude oil is not used in any significant way. The crude oil cools as it enters the ground facilities and it cools further as it is transported in the pipelines. No power is generated from the sensible part of the crude oil’s energy. Only the chemical energy is valuable upon combustion. The rest of the report relates to how prices are linked to the energy delivered.
From what has been discussed above, the thermodynamic analysis is incorrect and therefore any calculations and graphs based on this analysis must also be unreliable. Readers may note that the incorrect analysis predicts that one threshold based on their analysis was passed in 2012 and another will take place in 2022. That this coincides with the time others have judged to be when great difficulties to appear, seems to give the report a superficial credibility.
Since the chemical energy is the only significant energy of crude oil, this is the energy returned in the EROEI calculation. If the authors have a better handle on how much energy is expended in oil production, they can form the EROEI ratio and it would constitute an independent check on the work of Hall and his coworkers on EROEI. Such an independent analysis would be valuable.
430 responses to “On the Thermodynamic Model of Oil Extraction by the Hill’s Group”
Dr Turiel also analyzed this report and he found several problems with it.
Article, in Spanish, in his blog, The Oil Crash
Análisis de las bases teóricas del modelo ETP
http://crashoil.blogspot.co.uk/2017/02/analisis-de-las-bases-teoricas-del.html
Excellent articles by SK and Dr. Turiel. I like the conclusion from the crashoil blog (and a great job by Google Translate as well):
“That there is a need to have a model does not mean that any model is adequate, and I think it has been proven that the ETP model is not. However, the ETP model has been received quite uncritically by the community, in a collective error in which I myself have participated in some way. Possibly there has been a confirmation bias: as one colleague of mine, a bright and alert physicist said, the model started from the right premises and arrived at coherent conclusions, reasonably thinking that the model worked correctly. The truth is that very few had taken the trouble to calmly analyze the model and point out its shortcomings. In this sense, it is important to maintain a critical spirit and not accept without the things that seem to confirm what we believe; All the hypotheses must be examined and all the work must be reviewed, in order to obtain the best evidence, that provides the most useful results to everyone. Possibly, thanks to analyzes similar to the one I have presented here, The Hills Group can develop an improved model and with it we can improve our understanding of the dangerous road ahead.”
Except the Hills Group aren’t going to develop anything new and they (he?) are going to go on blithely ignoring all adverse comments.
So? No more “Fill ‘er up Joe”?
This is an excellent/balanced summary. Thank you to the author (and Dennis)
“Readers may note that the incorrect analysis predicts that one threshold based on their analysis was passed in 2012 and another will take place in 2022. That this coincides with the time others have judged to be when great difficulties to appear, seems to give the report a superficial credibility.”
Thats a perfect summation. THG report has some truths but is mostly appealing because of the catastrophe appeal. MIGHT end up being right but not for thermodynamic reasons. Modern society runs on oil, thus oil industry will be the absolute LAST industry to fail. It will be supported by hook or crook until then. Even at $200 a barrel we get energy a thousand times cheaper than human labor. Just not 20,000 times anymore.
Thank you for a succinct and clear analysis!
So, world oil production, over unknown time, approximates zero. Enlightening, Seriously, I get lost fairly easily.
Hi Guy M,
A layman’s guide.
Oil prices are not determined primarily by thermodynamics as the Hill’s Group suggests.
Geology and technology will affect the cost to supply the oil and World economic growth and technology will affect the demand for oil, the price of oil will mostly be determined by these factors along with policy and political choices made by individual nations.
Agree, but right now the price of oil is being determined by perception, only. It’s looney tunes time. The price of oil approached $100 far before the shale play. However, because there are a few places they can ply oil out of the shale at less than $40, then $55 is a fair price, because US oil can increase production by 3 million a day at $55 a barrel. EIA is estimating the US is producing about a half a million barrels a day higher than what my feeble mind can calculate that we are producing, so our supply must be in the stratosphere, somewhere. Somehow, Texas is supposed to keep up the extra 50 million barrels a month production the gained in the past five years with a very small fraction of activity that got them there, and turn around and increase that production with the same low level of activity. Why? Because someone read it on the internet, so it has to be true. Rig count went up three rigs, so the price of oil goes down, because we are swimming in oil, again. Time for the Tasmanian devil dance!
Time for the Tasmanian devil dance!
https://www.youtube.com/watch?v=3soT2pWboXU
You spin me right round Tasmanian devil dance
Good! D D Dats all Folks!
”Agree, but right now the price of oil is being determined by perception, only.”
NO. ABSOLUTELY NOT.
The price of oil is determined , broadly, over time, over the long term, by the total cost of delivering it to end users, and by the price end users are willing to pay for it.
In the short term, there are bed rock solid reasons why the price of oil may be up or down, well above or below the actual cost of production, and likewise rock solid reasons why end users will pay the prices they do actually pay.
Only idiots who have never actually read or been instructed in the theory of supply and demand think the theory calls for supply and demand to be precisely matched, and the price to reflect both , as in the nice illustrative graphs used in text books to introduce the concept.
In the REAL world, as it is explained by countless grad students and professors teaching intro to econ, in classrooms ranging from community colleges to the Ivies, the kids hear about how the price fluctuates and WHY it fluctuates.
Producers can never be sure how well their product will sell or at what price next year, never mind five or ten years down the road. Consumers can only guess at how much money they will have in future years, and how much they will be able to spend on oil, if they even bother to GUESS.
I have been involved in an industry just about my entire life that shares one key factor in common with the oil industry, that industry being the production of apples and peaches.
Lead times are long in both oil and orchards. Growers can only guess at what quantities will be coming to market five to seven years down the road, but they must make the decision THIS year, concerning new production down the road. A substantial part of the industry might be wiped out, as by a new blight or insect, as is happening to the citrus industry now. Or China might decide to wipe out Western domestic growers if free trade prevails, and the Chinese could do it, because their wage structure is peanuts, and their environmental regs are a joke.
Consumer tastes can change, spontaneously, or as the result of advertising, or the pronouncements of the health care industry, or demand for apples might crash or explode, depending on the state of consumer finances and the availability, or lack thereof, of substitute fruits and veggies, etc.
My family has been in the biz locally for a century, and we have seen prices so good we built very nice new brick houses and bought all new machinery for cash in a couple of years, and we have gone to town and worked out the losses for years at a stretch in textile and furniture plants before finally giving up and getting into another line, some of us, just before the price went UP again.
And note that orchardists are independent businessmen, and make decisions rationally, as best they can, based on their expectation of profits and losses.
You place your bets THIS YEAR for what you will be producing five to ten years down the road. IF fortune smiles, you win. IF fortune frowns, you lose your ass.
Now not only does the oil industry exist and work primarily on a similar five to ten year time frame, it is similarly at the mercy, but only to a minor extent, of the customer’s prosperity and preferences. The extent is minor because there are ( for now at least ) very few substitute products on the market. You can live fine without ANY apples, but oil is essential.
Now let us consider that MOST of the oil in the world is owned and produced by nationalized oil companies, and that governments are notoriously slow and inept at managing any sort of business, and that generating a profit might be and sometimes is secondary to other goals which can range from waging economic warfare with an enemy producer to maintaining a happy work force and smoothly functioning domestic economy so that the politicians in power can STAY in power. Happy voters vote for incumbents, and savvy dictators know that basically contented and secure vassals seldom revolt.
It’s commonly accepted except by liberals who will never admit that R types ever accomplished ANYTHING that the USA working mainly with the Saudi’s more or less broke the economic back of the old USSR by forcing the price of oil down back in the Reagan era for instance, and just about every body who knows doo doo from apple butter about international politics understands that there is and has been an economic war going on between various oil exporting countries for quite some time.
And anybody who has even the faintest conception of business realities understands that if you are IN an industry, lock stock and barrel, you can’t just QUIT when the price is unfavorable, you’re compelled to stick it out as best you can and hope you last until you’re in nice deep black ink again.
SO -IN THE SHORT TERM, and in the oil industry, short term means up to a DECADE or so, and the price can drop dramatically, or shoot up like a rocket, if the industry as a whole produces too much or too little, and it is to be EXPECTED that it will take a good long while for the cost of production to come into balance again with the price at which oil actually sells.
Then there is the whole entire concept of price elasticity to be taken into account. It takes only a little extra oil to glut the market and bring about a price crash, and only a minor shortfall to bring about a price spike, once the oil in storage starts piling up or running short.
Bottom line, as our fine host Ron Patterson has always said, the price of oil is determined by supply and demand, as is the price of virtually every commodity good, but in the case of oil, the industry necessarily moves like molasses in zero F weather, and it takes a few years for the oscillations to smooth out.
It would take many thousands of words to do a GOOD job explaining why the price of oil moves as it does and as slowly as it does, but there is nothing whatsoever wrong with supply and demand theory, when used as a TOOL by knowledgeable persons.
Now can the price of oil go as low as ten bucks or so, or even to zero? Well, I have seen the price of apples go so low it cost more to put them into containers and ship them to market than the delivered selling price, and we dumped them over a road bank into a gully to get rid of them. But independent oil producers can leave their oil in the ground when it is selling for less than the cost of actually producing it, unless compelled to produce to hold a lease, etc.
I repeat, GOVERNMENTS control the oil industry, and governments are only semi rational in terms of running the industry from the pov of a BUSINESSMAN.
But even after saying all this, and trying to explain why the price of oil may seem to be way out of reason, high OR low, I must come back and say that supply and demand determine the price of oil even in the short term. EVERY DROP that is produced, other than what is consumed internally by the industry, or put into storage by PRODUCERS, is SOLD, excepting a really trivial amount that is given away as foreign aid.
The price of oil could go to ten bucks, or zero, theoretically, but it wouldn’t stay there very long at all, because oil is an essential commodity, and oil will remain essential for decades to come, and the countries with the lowest production costs of them all still apparently have production costs of twenty bucks a barrel, according to what I have read.
And as the amount supplied increases, the price decreases, until end users are willing to BUY IT ALL. If the collective customer is NOT willing to buy it all, it STAYS IN THE GROUND,once the available storage capacity is all used up, because you CAN shut in an oil well or an entire oil field. You can’t shut down an orchard for a year or two hoping for a higher price later on.
Sure plenty of oil is produced at a loss. Lots of apples are produced at a loss too, but on average both industries are profitable.
If the price of gasoline were to fall back to fifty cents a gallon, I would drive my dump truck just like a car, because it would be cheaper to just do without a car, lol. If gasoline shoots up to six or eight bucks, I will make a point of thinking ahead so as to save some trips to town.
The very idea that some sort of cabal of business men or bankers or mafia or little green men controls the price of oil is ridiculous on the face of it. Why should a great white shart corporation such as EXON or SaudiAramco or Walmart or a giant chain of chain stores with gas pumps allow such hypothetical characters to get between themselves and their respective customer or supplier and rake off enormous profits at trivial costs?
The very IDEA is BULLSHIT.
When OPEC was functional, the cartel could influence the price of oil to a substantial extent, this is true, but OPEC consists of PRODUCERS, not shadowy parasites.
Who said anything about a cabal of nefarious individuals? Just the uninformed and lazy. Bullshit is reading additional information in sentences within the white parts of the paragraphs.
“t’s commonly accepted except by liberals who will never admit that R types ever accomplished ANYTHING that the USA working mainly with the Saudi’s more or less broke the economic back of the old USSR by forcing the price of oil down back in the Reagan era for instance,”
Oh, the Rs certainly did this, but it was an absolutely terrible idea and an evil thing to do.
Instead of allowing the humane, “glasnost and perestroikia” USSR of Gorbachev to evolve, the US decided to give total world power over to the Wahabbi extremist sheiks of Saudi Arabia who chop off the hands of bloggers and whip women for driving cars.
Republicans have most certainly accomplished a lot of extremely evil things.
Though Gorbachev himself says that the oil prices were not significant in the fall of the USSR. Most significant was the adminstrative mismanagement — everyone lying to their bosses because they were afraid to tell the truth — which caused the food shortages starting in the 1960s. He was trying to fix that. But the final deathstrokes were getting into a war in Afghanistan, “graveyard of empires” and bleeding the budget out on that, and having Chernobyl blow up.
The US seems to have repeated the Afghanistan war mistake exactly and precisely. All we need is our own Chernobyl/Fukushima event and it’s all over for the US. Hopefully we won’t have one, though (and the US will probably have a revolution anyway).
I think we’re having a revolution right now and I, for one, am scared silly because the revolutionaries are delusional, mean spirited and aggressive. A bad combination.
A real revolution has the peasants storming the gates of the wealthy and powerful.
This is a classic case of “revolution management”, in which the wealthy and powerful divert the peasants anger away from the real elite and towards scapegoats, like immigrants, environmentalists, intellectuals (the “cultural elite”) and government workers.
“The price of oil could go to ten bucks, or zero, theoretically, but it wouldn’t stay there very long at all, because oil is an essential commodity,”
No it isn’t.
” and oil will remain essential for decades to come, ”
No it won’t.
“and the countries with the lowest production costs of them all still apparently have production costs of twenty bucks a barrel, according to what I have read. ”
Yep… but that’s too expensive. The substitutes are cheaper even at $20/bbl. That’s the oil-industry-killing fact. Which I rub my hands together in glee about.
Now, existing wells can have *even lower* production costs (I’ve actually heard $2/bbl for older Saudi wells), so they’ll keep producing, but new well drilling will become utterly unprofitable. At some point it will stop, though I currently believe there will be a lot of really dumb unprofitable drilling first.
Hi Nathaniel,
If you think oil will not still be an essential commodity at least three or four decades down the road, you are either using a rather limited definition of essential, or one hell of a techno optimist.
The figure I used, twenty bucks, for low cost production, is one I took from someplace in my notes , that is commonly quoted as an AVERAGE production cost for countries such as the Saudis to produce oil. It’s the opinion of some industry people, sorry I can’t remember who. It may be possible that they do have some old wells that they can produce for as little as a couple of bucks a barrel, but inflation has been marching on for a long time now, and it costs a lot to maintain any sort of industrial infrastructure, so two bucks might be way on the low side.
Now we can play word games, and talk about the price of things being determined by perception, rather than reality, like children, some of us, but others of us have actually had shit on hand that MUST BE SOLD.
You may be an academic. The French have a saying, something to the effect that only a fool or an intellectual could possibly believe certain things.
Anybody that has EVER made his living selling stuff at wholesale understands that the retailers buy as much as they can sell, and that each and every business in the production and distribution chain spends as little as possible doing their part.
Gasoline could go to a nickel a gallon tomorrow, and the sale of it wouldn’t budge very much at all, for quite a while, because consumers want and can use ONLY SO MUCH.
Perception doesn’t have a goddamned thing to do with it. The end user buys as much as NECESSARY, or DESIRED, period, at the price he can get it. If the price goes low for a while for a commodity such as oil, people use more, everything else equal, because they buy bigger cars, boats, trucks, etc, but that takes a while. If the price of oil goes way up, they still buy almost as much, for a while, because it takes a while for the users to switch to more economical vehicles, or a smaller boat.
If the price of apples goes really low, some people buy twice, even three times as many, because they can substitute apples for other fruits and veggies, and if the price spikes, they buy half or a third as many, short term, and substitute other fresh fruits and veggies. The SUPPLY determines the price , more than any thing else, and producers ( Yours Truly) adjust supply to market conditions, as best we can.
You have read too many books about marketing. It’s probably true that perceptions determine the price of a lot of products, such as name branded beers, cosmetics, high status phones, etc.
It’s been proven many times that hardly anybody can tell the difference between the taste of hundred dollar booze and ten dollar booze in the dark, or the difference between the price of Pepsi and the Walmart brand soda over ice in the dark.
I have spent years and years riding in cars, and I can’t tell with a blindfold on whether I am riding in a forty thousand dollar Ford, or a hundred thousand dollar Mercedes Benz, except by feeling of the upholstery, running my hands over the door panel, etc.
It’s certainly unquestionably true that a Ford is about as reliable, and will last a LOT longer, considering that you can maintain a Ford for twenty or thirty years for the DIFFERENCE of sixty thousand in the purchase price. So perception does have a lot to do with the price of a Mercedes. You buy status with the extra sixty thousand, rather than transportation.
If the price of apples here in the USA drops to lets say five dollars a bushel wholesale, for two or three years, growers will QUIT planting new trees, on average, until the price gets back above production costs.REALITY will reduce the supply until the price is profitable again, given a little time. If it spikes to twenty bucks, and stays there a couple of years, orchardists will be buying new equipment, land, and hiring more help on the double.
Now you can say that these real world observations are based on perceptions if you please, it’s still somewhat of a free country.
And we now have an orange orangutan prez who assures us that alternative facts are as good as real ones.
But I want to remind my cyber buddy Fred Maygar that we ought to show more respect to our cousins the orangs, and I hereby apologize on behalf of both of us to any orangs who might be lurking here.
I will say that in the very short term, commodities spot prices are *always* determined primarily by perception. What else can they be determined by? All short term markets are like this, no exceptions. But that’s only the short term.
Buffett has been quoted as saying “In the short term, the stock market is a voting machine; in the long term, it is a weighing machine”. Same is true of commodities markets.
I loved the way Ron said it when refering to Bruno Verwimp’s chart,” The accuracy of his prediction is uncanny”.
The same can be said for the ETP Model. Who else so accurately predicted the reality since then, by Sept 2014 , ie falling crude prices, rising inventories. http://thehillsgroup.org/depletion2_022.htm
I find POB valuable(thanks to all you contributers) in keeping track of the numbers and progression of the oil industry, and those numbers keep confirming the ETP, just like it continues to verify Bruno’s chart applying Hubbert’s bell curve.
Nvertheless the whole concept is nonsense.
Non scientists have trouble understanding that a model or hypothesis might correctly describe what is going on and even in appearance predict it beforehand, yet be completely wrong. If Bruno Verwimp’s model and ETP model do not include all the relevant factors that we do know are part of oil production, then they are wrong no matter their present performance. End of the story as there is no way around that shortcoming.
There are multiple reasons why a model might appear to perform correctly, but only one why it doesn’t perform. One has to be aware of this asymmetry before jumping to conclusions.
It doesn’t take a high school education to clearly understand that correlation does not equate causation.
What many well educated people fail to understand is how critical net energy is. A high percentage of Americans can not even make the connection that if they would succeed in shutting down pipelines they would no longer have heating, electricity, or transportation.
POB has been busy counting barrels and $s but failed to figure in the net energy available to the rest of the economy. This kind of thinking led to expectations a couple of years ago, of crude prices in the hundreds of $ by now.
Saudi Arabia is a great example of producing ever more oil but exporting less using a big part of it just to produce the oil let alone refine it for the rest of the world. And they spend loads of their income to hire foreigners to do their work and to supply them with all the machinery they require. so in the meantime they are going broke spinning wheels, since the rest of the world can not afford to pay them what they need to make it work.
What better explains the current price of oil, given the massive quantities of money printing across the globe, and oil production flat for the last year and a half.
Until we have a better model that takes net energy into account, I choose to make decisions based on the ETP, besides it has an uncanny track record.
How many percent would net energy drop as EROEI declines from 15 to 10?
Yes, EROEI differences sound alarming the reality is not. 🙂
For 100 units of energy one gets
EROEI net energy
100 99
20 95
15 93
10 90
05 80
02 50
A decrease of EROEI from 15 to 10 gives a loss of net energy of ~3.5%.
As seen in every curve of EROEI ever published.
It falls off like a cliff as it gets near one.
Right now gasoline is around 2. Then only 20 percent of the energy in the gasoline gets to the wheels when used in an ICE.
It could be worse than that since a chunk of the energy input is electricity and steam, both of which are inefficient to produce from fossil fuel.
Hi Gone Fishing,
That EROEI for Solar PV looks pretty conservative, how old is that chart? My guess it is probably closer to where nuclear is on that chart as it is getting cheaper and cheaper.
Doesn’t matter, the point was the non-linearity of EROEI which the graph demonstrates perfectly.
Most EROEI calculations for PV are bogus anyway since they use a much shorter life for the panels than is probable.
There are already good, professional estimates of energy flows in economies, like the link below for UK.
https://www.gov.uk/government/uploads/system/uploads/attachment_data/file/539637/Energy_Flow_Chart.pdf
Hi Farmboy,
I agree net energy is important, but only as it relates to all sources of energy used by society. When the ETP model looks at energy flows into and out of the petroleum production system (PPS) it needs to account for the source of energy, in many cases the energy is supplied by coal or natural gas and not by oil.
Simply put it is the net energy of all forms of energy which society uses that is important, not the net energy of a single product (oil).
What is the net energy of the electricity you use? Does it affect the price you pay? My guess is that you have no idea what it actually is. Or maybe you do, most people have no idea, so it has little bearing on the price they pay.
The companies that produce the electricity also don’t bother to find the EROEI of the electricity produced, the point is to supply the electricity at as low a cost as possible to keep profit margins as high as possible at the market price.
The maths and physics is above my pay scale , but they came at two conclusions
1. The price of oil is going to go down and devolped a maximum affordability curve for future price levels .
2. In the future there will be a glut of oil .
I agree with Ron´s statement made by Farmboy in his post . The point is that both conclusion have stood well for 2015,2016 and the first 2 months of 2017 . Is this a fluke ? I don´t know ,but if yes this surely a nugget. Sometime you arrive at the correct conclusion just by chance ,ask Louis Pasteur .
The proof of the pudding lies in the eating .So far so good .
A lot of problems with the analysis besides just the thermodynamics.
A basic assumption of the theory is that oil has to be a source of energy. Not clear why.
As long as people need oil they will pay a price for it, the price has to be high enough so that demand can be met by suppliers at a profit (in the long run). It really is that simple. Just as we are willing to pay for electricity, we would be willing to pay for oil even if it’s net energy was negative. It is the net energy of all social energy sources that is important, not that of a single product.
Dennis said: >> we would be willing to pay for oil even if it’s net energy was negative <<
True. Take AA batteries. It takes 381 Wh to produce a battery providing 4.2 Wh of energy. Hugely net negative. We buy them buy the billion…
Nobody uses AA batteries as a prime mover. People buy the batteries for their convenience.
The next question is ‘what is pay’?
Money is just a promise to borrow again later. That (empty) promise cannot put oil in the ground, in fact, nothing can. If the amount of work needed to lift oil is greater than the work that can be done with the oil the enterprise breaks down. At present the money system is unraveling due to the need for borrowings that are greater than what any number of promises can deliver. There is not enough borrowing and the oil remains in the ground because it is unaffordable.
Basically, this is what Exxon said in not so many words the other day when it wrote off 15% of its reserves.
Nobody uses AA batteries as a prime mover. People buy the batteries for their convenience.
True but lithium-ion batteries are a whole nuther can of worms.
Check out both Jack Rickard’s last month’s and his most recent video ” Hacking the Tesla Battery Module”.
http://evtv.me/
See also: ‘3 More Gigafactories Coming Soon to ‘Change the Way the World Uses Energy’
http://www.ecowatch.com/new-tesla-gigafactory-2280389543.html
The company said in its fourth-quarter investor letter on Wednesday that it is considering building up to five Gigafactories.
Tesla is far from the only game in town… there are ten other major battery companies with plans as well.
Debt is the prime mover of energy prices. When the world was loading up on debt, the price of Oil soared. It appears that the world is at or near debt saturation, and adding further debt likely decreases economy activity because the costs to service the debt suck capital out of the real economy. Paying down debt also decrease economic actively. I don’t think we will see Oil prices move significantly higher (excluding a major geopolitical event).
Looks like the Pension bomb is beginning to explode this year. This is likely to cause severe havoc as pension after pension goes bust or announce heavy benefit cuts.
NY Teamsters’ Pension Fund Goes Belly Up
http://freebeacon.com/issues/ny-teamsters-pension-fund-goes-belly/
Californians Hit as Bad Debts Lead to Government Pension Cuts
https://www.bloomberg.com/news/articles/2017-03-01/californians-hit-as-bad-debts-lead-to-government-pension-cuts&rct=j&q=&esrc=s&sa=U&ved=0ahUKEwi-g_S507nSAhVM54MKHZUvCO4QqQIIFCgAMAA&sig2=8R5TeQkBu2DcQORuDzUt7w&usg=AFQjCNEZnnfWOQ0cM_PQO5aPYeS4QZZnBA
America’s utterly predictable tsunami of pension problems
https://www.washingtonpost.com/opinions/americas-utterly-predictable-tsunami-of-pension-problems/2017/02/22/1e5de00e-f869-11e6-9845-576c69081518_story.html
CMBS Delinquency Rate on the Rise
http://nationalmortgageprofessional.com/news/62261/cmbs-delinquency-rate-rise
$1 Trillion Car-Loan Market Has Highest Delinquency Rate Since 2009
http://www.investors.com/news/car-loan-delinquencies-hit-highest-level-since-2009/
Fannie Mae’s Serious Delinquency Rate Decreases to 1.20%
http://www.builderonline.com/money/mortgage-finance/fannie-maes-serious-delinquency-rate-decreases-to-120_c
[More retailers are closing stores]
JC Penney Closing 140 Stores in Early 2017
http://ww2.cfo.com/budgeting/2017/02/jc-penney-closing-140-stores/
HHGregg closing 88 stores amid bankruptcy rumors
http://www.indystar.com/story/money/2017/03/02/hhgregg-closing-88-stores-amid-bankruptcy-rumors/98629544/
Macy’s committed to closing 34 more stores as sales drop in 2016
http://www.nj.com/news/index.ssf/2017/02/macys_committed_to_closing_34_more_stores_as_sales.html
Sears Holdings unveils restructuring plan amid 150 store closures
http://www.al.com/business/index.ssf/2017/02/sears_holdings_unveils_restruc.html
Not sure if Oil prices will dip much, but fuel prices for consumers will likely be increasing as many states are increasing their gas taxes for 2017. With the Shale busts, its likely that production will fall and offset consumption declines.
Oil Patch Bankruptcy Total Rises To $110 Billion
http://www.valuewalk.com/2017/02/oil-patch-bankruptices/
My guess this there will be about $60B to $120B additional bankruptcy/write downs in 2017. Interest rates are going up and its going to put a squeeze on struggle businesses & consumers.
Odds are that the US will be back in recession in 2H of 2017 or by 2018.
The stock market makes no sense to me. I don’t see anything on the horizon that will boost the economy and the latest GDP figures weren’t impressive.
America’s gonna be sooo GREAT again! Can you say schadenfreude?!
How are internet sales doing?
From what I see retail in general is rising yoy. Certain stores may be stuck in descending or static areas.
https://www.census.gov/retail/mrts/www/data/pdf/ec_current.pdf
What’s interesting about that graph is how the adjusted dotted line levels off mid 2016, just like ~ 2008.
However, most of exxon’s write off will go back on the books as oil prices increase. The only loss will be wells abandoned because they are too far below the economic limit.
I have been in similar circumstances, and saw the reserves go back in once prices recovered. The key in some cases is to finagle well stock so they can be opened again. In some cases the completion equipment and the pumping unit justify pulling the well. It takes a lot of thought to optimize what to do during price swings.
The only problem with your argument: Oil prices won’t recover. They have exactly one chance to recover, between now and 2023 (and that’s a generous assumption), and then they crash for good. This is easily computable from the demand side.
If the irrational exuberance we see in the oil business continues, they won’t even get that one last dead cat bounce of high prices.
Hi Fernando,
Did you look at the Turiel blog post?
My Spanish es muy mal, y no entiendo bien.
I tried Google translate and that gave a bit of a feel, but your take would be interesting as I imagine you have taken more than your share of thermo and pchem.
“If the amount of work needed to lift oil is greater than the work that can be done with the oil the enterprise breaks down.”
~steve from virginia
Steve is absolutely correct on this.
If the mass (oil and water) and heat removed from a control volume (oil reservoir) are known, then the Entropy Rate Balance Equation For Control Volumes can be used to calculate the exact amount of energy expended in the process. Simple. This is the Energy Invested (EI) part of the ERoEI of oil.
The chemical energy of the oil, released through it’s combustion, is the Energy Returned (ER).
It is pretty obvious that most people here simply don’t understand even the most basic concepts of physics, or the Etp model, at all. The article by SK is total gibberish. Amazing.
Out of interest what do you think about the spanish article linked in the first comment above, also by a highly qualified and experienced academic who specialises in thermodynamics?
ERoEI is irrelevant. As Rockman (on peakoil.com) often points out, it’s costs that matter. Production costs versus selling price. Nobody in the oil patch does ERoEI analysis.
It costs less to produce an AA battery than its market price. Even though it takes approx 100 times more energy to produce it than is contained in the battery.
[60 MJ/kg; see PDF at http://bit.ly/2lGxRP4%5D
That’s probably a better explanation for our current system than energy efficiency. As long as someone is willing to pay people to drill (by either buying the stuff or subsidizing it or being willing to take a loss) people will drill. It doesn’t have to make economic sense for the economy.
We are very likely to reach the limits of people’s willingness to pay others to drill. They will find substitutes or do without if the price is more than they want or can pay.
Is that you Loren Soman?
Hi Cunning linguist,
So you think the mechanical engineering professor does not understand classical thermodynamics. You are ignoring the irreversibilities in the extraction of oil, the mechanical engineers are pretty well versed in mechanics and thermodynamics, perhaps it is you who needs a refresher on your basic physics?
Note that you are assuming the Hill’s Group has made correct assumptions in their thermodynamic analysis, they have not.
There is a good reason the ETP model has not been published in a peer reviewed journal, it would likely be rejected by qualified reviewers.
‘The mechanical engineering professor’? Is that, like, The Pope or The Queen?
Has this person, their specialization, research and place of research been verified?
If so, by whom? You?
So we’re supposed to take your word for it?
What about peers?
What peers, who peers, where peers?
So I guess the ‘encroaching model’ of science, engineering and/or peer-review, etc., can be undertaken and reviewed by a whole bunch of anonymous entities?
So, say, if anything goes wrong– not that when things related to science and engineering go wrong it’s all that important mind you– can we slowly forget about accounting or accountability and/or things along those lines?
Here we go gathering nuts in may
‘The mechanical engineering professor’? Is that, like, The Pope or The Queen?
Has this person, their specialization, research and place of research been verified?
Most universities do verify the credentials of their faculty, so if he is a professor at a university he probably has a pretty good grasp pf thermodynamics…
Typical curriculum for MS in mechanical engineering…
http://engineering.nyu.edu/academics/programs/mechanical-engineering-ms/curriculum
That’s irrelevant, or, at best, tangential, to my point.
So what exactly is your point, Caelan?
That I shouldn’t trust Dennis’ judgement with regards his choice of who he allows to post on his and Ron’s blog?
While I don’t know who SK is I certainly accept that Dennis does and have no reason to doubt his credentials or competence. Based on all my previous interactions with Dennis I have no reason to doubt his understanding of basic physics and thermodynamics.
I may disagree with Dennis in terms of degree on certain topics but not in this case. I have followed the Hills group ETP model discussion for a long time. I’m convinced it has little merit. Are you trying to defend it? If so on what basis?
Dear Fred,
You said: “I’m convinced it has little merit. Are you trying to defend it? If so on what basis?”
Well, I am convinced that the Etp model has great merit, and I have already explained why. Are you trying to debunk it? If so, on what basis?
Seriously man, do you just take Dennis’ (and SK’s) word on all this? Is that your only reason for thinking the Etp model is invalid? I hope not.
If you can think for yourself, please try to put in your own words why you don’t trust the Etp model or it’s forecast. Thanks.
The first time he posted he used his full name. I looked him up and he has an impressive resume. The spanish paper cited above repeats pretty much what SKs analysis says. Almost any reasonably educated postgraduate in the thermodynamics field could do the same if they could be bothered, it is not cutting edge research. But why waste their time – the Etp theory is utter tripe from beginning to end.
Correct!
Otro de los problemas conceptuales de partida es que todo el análisis toma el PPS aislado del resto de la economía y en particular de otras fuentes de energía que podrían apoyar la extracción de petróleo (sustancia que podría seguir siendo interesante por su mayor valor añadido), y que en todo caso hace que las afirmaciones sobre el colapso del PPS y de toda la sociedad sean como mínimo cuestionables: puede ser que tal cosa vaya a pasar, pero no es lógicamente ineludible a partir de lo que se está analizando teóricamente.
As they say, even a blind squirrel occasionally finds a nut! Cum hoc ergo propter hoc….
Hello Dennis. You said: “So you think the mechanical engineering professor does not understand classical thermodynamics.”
1. You are tying to make an argument from authority. This has no validity.
2. I do not suggest that the engineering professor does not understand basic classical thermodynamics. Engineers learn about closed (reversible) systems before they study open (irreversible) systems. Most never even get around to studying open systems at all, as this is more generally the domain of ecologists. This results in engineers who, in this case, overemphasize what they are most familiar with, i.e. the study of idealized reversible processes found in machines.
In natural systems, the entropy production rate of every process is always positive (ΔS > 0) or zero (ΔS = 0). But only idealized adiabatic (perfectly insulated) and isentropic (frictionless, non-viscous, pressure-volume work only) processes actually have an entropy production rate of zero. Heat is produced, but not entropy. In nature, this ideal can only be an approximation, because it requires an infinite amount of time and no dissipation.
An irreversible process degrades the performance of a thermodynamic system, and results in entropy production. Thus, irreversible processes have an entropy production rate greater than zero (ΔS > 0), and that is really what the second law is all about (beyond the second law analysis of machines or devices). Every naturally occurring process, whether adiabatic or not, is irreversible (ΔS > 0), since friction and viscosity are always present.
SK wants to define and study the specific mechanical causes of the irreversibilities that build in the oil production process. This is admirable, but not necessary. The Entropy Rate Balance Equation already accounts for all of the irreversibilities and simply gives the total entropy production of the system being analyzed.
You said: “Note that you are assuming the Hill’s Group has made correct assumptions in their thermodynamic analysis, they have not.”
You certainly have no way to evaluate whether the Hill’s Group is correct or not. I am not assuming anything. I have a good physics background and I spent more than a year studying the Etp model and all of the arguments made against it. SK’s article is just a rehash of the usual lame, invalid objections dressed up in fancy physics language to make it seem authoritative. You don’t understand the Etp model or SK’s misleading article. Yet you are sure the Etp model is wrong. This is funny.
I also have a good physics background having majored in it as an undergraduate. I will go with the evaluation of the Professor emeritus. There are several people who post here with good physics backgrounds who are unconvinced by the ETP model.
Dennis, you said: “I will go with the evaluation of the Professor emeritus.”
Argument from authority.
The Professor emeritus might understand thermodynamics, but he still shares the same misunderstanding of the Etp model that I just corrected Rune on (down the page). SK spends most of the article arguing that heat dissipates in pipes on the way to consumers! This has nothing to do with the model at all and, in fact, it just highlights his basic misunderstanding of it. SK’s critique is a straw man.
Hi CL,
You have not understood what was written.
The irreversibilities are when the oil is extracted. Reread the post carefully. He said very little about losses after the oil is extracted. A basic assumption of Hill’s analysis is incorrect, see the second equation in the post, incorrect assumptions lead to a flawed analysis. I noticed so far Hill’s attempts at rebuttal essentially are name calling. Yours, argument from authority.
I believe SK’s analysis both because he is an authority and because he is correct.
Cunning wrote;
”I have a good physics background and I spent more than a year studying the Etp model and all of the arguments made against it. SK’s article is just a rehash of the usual lame, invalid objections dressed up in fancy physics language to make it seem authoritative.”
Since you are such an expert on the Etp model, could you please reference the equation where chemical energy of oil and gas is described?
Rune, you said: “Since you are such an expert on the Etp model, could you please reference the equation where chemical energy of oil and gas is described?”
???
Your question makes no sense. The chemical energy contained in a barrel of oil is constant and well known. The Etp model does not use an equation to derive it, so there is not one described in the report. The absence of such an equation is not a problem for the model. It is only a problem for you. It exists in your mind only, because you still don’t understand how the model works. You keep looking for it because you think the Etp function has to be somehow based on the chemical energy of the oil. It doesn’t. The Etp function is derived from the Entropy Rate Balance Equation for Control Volumes, using the mass and heat removed from the reservoir as inputs. You will just have to accept that even if you can’t understand why it is true.
Somehow the Etp model needs to describe the flow of oil and gas, so even if you find it hard try to be a little helpful.
Hi Cunning Linguist,
I guess I will let the professor speak for himself. There was not an argument just a question.
So I will ask another “argument” :), Do you think a mechanical engineering professor is not familiar with open systems where there are irreversibilities?
Yes Hill is ignoring those, the point is that the professor is not.
Entropy of the universe always increases, depending upon the boundries of an open system and the heat flows, it is not always true that the change in entropy will be positive.
Hi Dennis.
You just boldly/ignorantly claimed: “Entropy of the universe always increases, depending upon the boundries of an open system and the heat flows, it is not always true that the change in entropy will be positive.”
Please name an open system in which entropy production is negative (ΔS<0)!!!
You can’t, because everywhere in the universe, entropy production is either positive or zero. There are no exceptions to this.
Please read this again:
"In natural systems, the entropy production rate of every process is always positive (ΔS>0) or zero (ΔS=0). But only idealized adiabatic (perfectly insulated) and isentropic (frictionless, non-viscous, pressure-volume work only) processes actually have an entropy production rate of zero. Heat is produced, but not entropy. In nature, this ideal can only be an approximation, because it requires an infinite amount of time and no dissipation.
An irreversible process degrades the performance of a thermodynamic system, and results in entropy production. Thus, irreversible processes have an entropy production rate greater than zero (ΔS>0), and that is really what the second law is all about (beyond the second law analysis of machines or devices). Every naturally occurring process, whether adiabatic or not, is irreversible (ΔS > 0), since friction and viscosity are always present."
Are you seriously disputing any of that?
Are you claiming that the oil production process is reversible? LOL!!!
Hi Cunning Linguist,
What you say of entropy is only true for an isolated system, it does not apply to an open system where there are flows of mass and heat across the system boundries. It is true for the universe, but not for all systems where depending on the boundries and flows of heat and mass, entropy can increase or it can decrease.
See https://en.wikipedia.org/wiki/Entropy
The second law of thermodynamics states that an isolated system’s entropy never decreases. Such systems spontaneously evolve towards thermodynamic equilibrium, the state with maximum entropy. Non-isolated systems may lose entropy, provided their environment’s entropy increases by at least that amount.
You have assumed all natural systems are isolated systems, where for the natural system of interest (planet Earth) there are flows of heat and mass across the system boundries, that is we have an open system not an isolated system.
For the universe, I would agree with your assertions and entropy would indeed always increase for an isolated system since irreversibilities are never zero in the real world.
Not that Hill’s assumptions, require that we assume irreversibilities are zero for the proposed equations to be true.
You just don’t see this because the assumption is not made explicit.
So it is Hill, not SK assuming irreversibilities are zero. Note that Hill has not addressed this, but prefers name-calling.
Hi Cunning Linguist,
You asked:
Please name an open system in which entropy production is negative (ΔS<0)!!!
From https://en.wikipedia.org/wiki/Entropy
see (“Entropy of a system” link)
As an example, for a glass of ice water in air at room temperature, the difference in temperature between a warm room (the surroundings) and cold glass of ice and water (the system and not part of the room), begins to be equalized as portions of the thermal energy from the warm surroundings spread to the cooler system of ice and water. Over time the temperature of the glass and its contents and the temperature of the room become equal. The entropy of the room has decreased as some of its energy has been dispersed to the ice and water. However, as calculated in the example, the entropy of the system of ice and water has increased more than the entropy of the surrounding room has decreased. In an isolated system such as the room and ice water taken together, the dispersal of energy from warmer to cooler always results in a net increase in entropy.
So take a similar example, but instead of a glass of ice water we take a pot of boiling water with a lid and turn off the stove.
In this case as the water and pot at 100 Celcius (the system) comes to equilibrium with its surroundings at room temperature, the entropy of the hot water and pot will decrease as the heat flows from the hotter object to the cooler surroundings the change in entropy of the system (water, pot, and lid) will be negative because heat flowing from a hot object to cooler surroundings is a heat loss (Q is a negative number). Is the second law of thermodynamics violated? Of course not, the entropy of the surroundings increases by at least as much as the loss of entropy of the hot water, pot, and lid, as the process is not reversible the entropy gain by the surroundings will be more than the entropy loss by the cooling system.
The fact remains that the system’s entropy has decreased.
Hi Steve from Virginia,
Is oil the only source of energy used to lift the oil from the ground?
No. Most of the energy for lifting oil is provided by electricity produced with coal, natural gas, hydro, nuclear, wind, and solar power.
Do you pay for electicity? Or is the price zero where you live?
What is the EROEI of the electricity you purchase, assuming you use electricity and its price is not zero?
Let’s assume you use electricity and your price is non-zero. Why is it not possible that oil might not be an energy carrier in the future (like electricity is today)?
Also not that I am not claiming that oil is an energy carrier, Rune Likvern has given numerous examples showing that for Statoil (not usually considered the lowest cost producer) the oil produced is nowhere near being net energy negative. My recollection is that it is at least an EROEI of 10:1 for Statoil, but Rune can correct me if I have remembered incorrectly.
Dennis, you said: “Is oil the only source of energy used to lift the oil from the ground?”
Your question is not valid. It shows your general ignorance of the model you are attempting to critique. The Etp model takes into account ALL forms of energy used to produce, refine, and distribute oil and it’s products. Some of the energy comes from oil, some from solar, some from wind, etc. The Entropy Rate Balance Equation can only output the sum TOTAL energy used, it does not give a separate tally of each source. I hope this helps clear things up a bit.
Entropy rate balance equation is used improperly by the ETP models where incorrect assumptions are used.
A claim that all assumptions are correct does not make it so.
Irreversibilities, sigma in the rate balance equation, are assumed to be equal to the heat flow out of the oil reservoir.
This assumption needs to be proven. There is no a priori reason why this would be true. You accept this because the Hill’s group makes the claim. They make many claims which are not believable. Chief among them is the proposition that the price of oil is related to its net exergy.
It sounds nice, but in general no other good’s price is determined in this way, the premise is false.
Dennis, you just said: “Irreversibilities, sigma in the rate balance equation, are assumed to be equal to the heat flow out of the oil reservoir.”
No, they aren’t. You are once again showing that you have no understanding of thermodynamics or the Etp model. You are making a false claim about how the model works. It is just a straw man argument.
The Entropy Rate Balance Equation for Control Volumes calculates how much entropy is produced in a control volume when MASS and heat are removed.
You also said: “This assumption needs to be proven.”
That is totally ridiculous, Dennis! You are treating the second law of thermodynamics as an assumption that needs proving. What a joke! Please stop embarrassing yourself.
You also said: “Hill’s group makes the claim. They make many claims which are not believable. Chief among them is the proposition that the price of oil is related to its net exergy.
It sounds nice, but in general no other good’s price is determined in this way, the premise is false.”
You say the premise is false, yet the Etp model continues to correctly forecast the declining price of oil as well as rising inventories.
The Etp model deals with oil. Oil is not like any other good. Oil is primarily an energy source. It’s use in the economy has to generate enough GDP to pay for it’s continuing production. The extremely high correlation between oil production and world GDP is not a premise or an assumption. It is a fact. Do you understand?
Hi Cunning Linguist,
Clearly you don’t understand the post.
I am not embarrassed at all, the ETP model suggests that using the first equation in the post, that one must make the assumption that the second equation in the post is true at all times in order for the third equation to be true.
One only needs to know basic algebra to see that this is true.
Problem is the second equation is not true at all times if there are irreversibilities in the extraction of oil.
Very straightforward, but clearly you do not understand.
But insult me some more as that definitely shows how much you know.
Hi Futilitist,
That is a clever rhetorical device trying to make it look like I said the second law of thermodynamics needs to be proven.
Clearly not what I said, the assumption made by Hill to get from the first equation of the post to the third equation in the post (which is correct, Hill has this wrong) is the second equation in the post.
That assumption needs to be proven, it is below and must be assumed during the entire oil extraction process for equation 3 in the post to be true.
Oil is primarily a handy way to store energy, wich is why we use it. As a source of energy it is too expensive.
Steve,
What is your point as globally we are now far from a point where the energy/work provided from oil and gas is less than the work/energy required to make it available for consumers.
There are several studies in peered reviewed papers that put the EROI at 8 – 40 for oil and gas (and it has been in a general decline). All serious (as in quality checked studies by professionals) shows the same range.
One study shows trendline(s) that brings global EROI for oil/gas to about 15 now.
If EROI drops from 14 to 9 it does not constitute a disaster (it is manageable) and such a drop could take years/decades to play out.
”If the amount of work needed to lift oil is greater than the work that can be done with the oil the enterprise breaks down”
This is not necessarily true. So long as oil can be extracted, processed, and delivered at a price that the end user can pay, it will be.
And the energy used to do the extracting, processing, and delivery does not NECESSARILY have to come from oil. In actual fact, not all of it DOES come from oil. A fair amount comes from natural gas, which is close enough to call it oil maybe, and some comes from coal, some comes from nukes, some comes from hydro, and some even comes from wind and solar power.
It wouldn’t matter a tinker’s damn if the amount of natural gas sourced energy used to produce tar sands oil is five times the energy content of the oil, so long as the natural gas used to provide that energy is CHEAP ENOUGH and PLENTIFUL, and so long as there is a market for tar sands oil that earns a profit above the monetary cost of producing it. (I am ignoring externalized environmental costs for simplicity’s sake in making this comment. )
EROEI is a very useful concept, a critical concept in fact, but it only applies as a practical matter once ALL sources of energy become expensive, or when the source of energy used to run a certain industry becomes expensive. So long as gas is dirt cheap in Canada, and oil sells for ENOUGH money, the tar sand industry will be ok, financially and economically.
Why ANYBODY has ANY trouble understanding something so simple totally escapes me. Some things are problems only at he margins. EROEI is one of those things.
We use several times as much energy (a factor of ten is commonly quoted ) producing food as food itself contains.
Even if we go back to farming with draft animals, the EROEI on grass fed to horses won’t matter, so long as grass is plentiful and cheap. We naked apes can’t eat grass, as such, but we can surely eat corn and wheat, etc, and it’s the energy content of the corn and wheat that MATTERS. The energy content of the grass eaten by the plow horse DOES NOT MATTER – so long as there’s an ample supply of grass.
And the energy used to produce grass, once the pasture is established, is essentially free and unlimited, at least so long as the sun continues to shine, lol.
At some point oil field operators in places with lots of sunny weather will use pv systems to drive some of their machinery, because such systems will be cheaper to own and operate than diesel or gas fired generators, or extending power lines to the wells. This has in fact been done already unless I’m mistaken.
“At some point oil field operators in places with lots of sunny weather will use pv systems to drive some of their machinery,”
Dubai and Abu Dhabi are already doing this routinely. (The UAE are way more forward-looking than the rest of the oil countries.)
Have a Source for the Wh to produce a 14500 “AA” disposable Battery?
I just presented a fun battery economics dog and pony show for a local IEEE meeting. Energy input to make a “Tesla” Panasonic NCR 18650B ( 3.7V 3400mAh ) ?? – it’s quite a complicated recipe ( Li, Mg, Co, + Al ) and production process
LT, yes, derived from a 60MJ/kg number from this PDF:
http://bit.ly/2lGxRP4
PS What was your number for the 18650B?
Excellent analogy. We use liquid fuel because it is a great way to store energy. As an energy source it can’t compete, which is why it is not popular as a way to generate electricity.
Even cheap oil can’t compete with coal, gas, nuclear and renewables as a source of energy.
That is why the continuing collapse in battery prices is such a big deal for the oil business.
“We use liquid fuel because it is a great way to store energy.”
That doesn’t sound right?? You don’t need much to store a pile of coal, but you pretty much need a tank to store liquid.
Liquids are easier to transport, separate, process, inject, modify, etc.
Exactly – oil is a very convenient way to store, transport and use energy for certain things: basically, things that move.
Oil lost out against electricity long ago for interior lighting; it lost out a while ago for electrical generation for large, well run grids; and now it’s losing out for passenger transportation.
It will hang on for a while for certain things, like seasonal ag, long distance land and water transportation and aviation.
Eventually, of course, even these will eliminated, whether it’s by better batteries or synthetic liquid fuels.
Oil gives us “little” more energy than batteries. Batteries are subsidied by other energy sources, but what energy source will subsidy 35% of world energy consumption?
“Oil gives us “little” more energy than batteries. Batteries are subsidied by other energy sources, but what energy source will subsidy 35% of world energy consumption?”
First off, I have pointed out in other comments that I can saw twice as much wood, easily, in a day using my ICE powered chainsaw as I can with the most powerful available battery operated saw, and it will run all day on a gallon of gasoline, cutting firewood, whereas the electric saw would require at least eight hundred to a thousand dollars worth of spare batteries to take it to the woods all day using it the same way. Oil provides more than a LITTLE energy compared to batteries.
AND
We have plenty of reason, having observed other stars by the millions and billions ever since the invention of the astronomical telescope, to believe that OUR star will continue to perform reliably for at least another billion years or so. It provides thousands of times more energy than we are ever likely to want, and we know how to capture that energy at fast decreasing cost.
If we last so long, we can probably figure something out in a billion years. 😉
The real question is whether we are smart enough to keep the pedal to the metal and push the growth of the renewable energy industries to the max, because time is short, and pushing very hard may well be the difference between a successful transition and a crash and burn economic and ecological collapse due to the one two punch of fossil fuel depletion and environmental degradation.
I hope and pray that Fred Maygar and other techno optimists are right about how fast these technologies will grow, and that such new technologies will be born and grow up and displace fossil fuels even if we DON’T force feed them like pa te geese.
But the precautionary principle is one all wise men and women hold dear, at all times, and this particular case is the most important one of all time.
BESIDES……….
I can’t prove it, but I think maybe we are already getting back a net profit, society wide, in terms of dollars alone, on our investment in renewable subsidies , due to wind and solar power ALREADY putting powerful downward pressure on the quantities of coal and gas used, and minor pressure ALREADY on the quantity of oil used.
When you reduce the quantity used of a commodity good, you also reduce the PRICE of it. So not only are we using less fossil fuel already as the result of subsidizing wind and solar power, we are getting a price break as a BONUS on the coal, gas , and oil we CONTINUE to use.
If anybody comes across any figures produced by people or organizations with the expertise to explore this possibility, please post links here and thanks in advance.
And incidentally, if you want D’s to win elections, when you happen to come face to face with a living cave man aka a REPUBLICAN, talk about driving down the cost of fossil fuels rather than climate change, and you will get his attention, and plant the seed of subversion in the soil between his ears, where it will sometimes grow like crabgrass in hot wet weather.
That sort of man does not TRUST renewable energy, but you can take it to the bank that neither does he harbor any affection in his heart for his electric utility or the oil industry, unless he OWNS stock in them. THAT sort of man likes to have as much personal control over his life as possible, and he will be installing a pv system of his own more and more often, and buying an electric car as soon as the price of them comes down enough to justify the higher up front cost of them, and he is confident about the resale value of them, etc.
Incidentally the early adopters of electric cars among men of that sort aren’t much in favor of subsidies, although they are quick to take advantage of them.
They generally remember a little detail referred to as OPPORTUNITY COST, which is THE detail that determines my personal decision to only own and drive old cars. Over the years I have INVESTED the money my acquaintances spent on new cars, and the investments had a hell of a lot to do with freeing me from the need to WORK on a regular basis at an ordinary career.
Thanks for confirming that posters here on POB are clueless
Welcome to the POB, Asdf.
We await your leading us to enlightenment with bated breath.
Hi Asdf,
Sun, wind (based on solar energy), hydro (also based primarily on solar input), so basically the sun, which is also the original source for most of the energy in oil, coal, and natural gas.
So the sun, same as it ever was.
Keep in mind that roughly 35% of primary energy consumed is oil at roughly 20% efficiency (when all aspects of the petroleum production system and thermal losses are included). So from an exergy point of view only one fifth of that primary energy needs to be replaced (if a 100% efficient energy source replaced it), for EVs, it is likely that the system would be at least 80% efficiency, so we might only need 25% of the primary energy provided by oil. Note that oil output will not fall to zero overnight so there will be some time to accomplish such a transition (probably about 40 years or so, though it might happen more quickly, it will depend in part on the relative price of oil and electricity in terms of exergy per dollar spent).
Dennis Willing to pay for oil does not equate ability to do so. Growing up dirt poor has ingrained that quite well.
Until someone developes a source of energy significantly better than oil, the worlds economy will continue to contract until it is only a shadow of its former self.
Hi farmboy,
Oil will become more expensive as it becomes scarce and we will use it more efficiently and we will also substitute EVs and public transport in some cases.
”Oil will become more expensive as it becomes scarce and we will use it more efficiently and we will also substitute EVs and public transport in some cases.”
Precisely. So long as the supply declines only gradually, giving us time to adapt, the world will be able to run just fine on oil that costs well over a hundred dollars a barrel in present day money, or even two hundred dollars a barrel. The energy intensity of the overall economy has been declining for some time, and it will continue to decline for the foreseeable future.
Workers who earn their living cutting hair or flipping hamburgers will be replaced by workers building and or refurbishing houses to use half or even a quarter as much energy.
Even here in the backwoods of the boonies, I already know a couple of people who are driving electric cars and making a substantial part of their own electricity. If things go as planned, I will be installing my second piece of solar powered machinery this summer.
And a ratted out LEAF or VOLT car when the ancient Buick in the driveway finally needs a major repair is in the cards. A Volt would enable me to cut my personal ( non business related ) oil consumption by at least eighty percent. I like wood heat, and use an existing high efficiency oil furnace as back up, burning seventy five to a hundred plus gallons a year in the furnace. When the day comes I am no longer able to ENJOY harvesting and using wood, I will put in a heat pump, which will just about wipe out my personal oil consumption. The lawn mower only uses about ten gallons a YEAR, lol.
One truck loaded with potatoes can deliver ten times as much food as a truck loaded with potato chips, lol, and the bags used for potatoes are potentially reusable.
Slow down from sixty to thirty five, and you can come close to doubling fuel economy without doing anything else at all, truck or car. You can safely haul a heavier load by going slower, which accounts for part of the savings.
I haven’t looked for figures, but there is little doubt in my mind that commercial farmers are improving their overall energy efficiency by at least one or two percent a year, and there are technologies already in use, although not yet widely adopted, that will enable even greater improvements.
the worlds economy will continue to contract
That’s fascinating. What gives you the idea that it’s contracting now??
Resource draw-down, environmental degradation, increasing wealth disparity and social unrest, etc.?
Thanks to SK for taking the time for this detailed and excellent review of the Etp report and filling in the missing links and put the findings in writing.
Also, thanks to Dennis for posting it.
There is an unbroken rule within engineering and science;
A report should be structured and written in such a way that any competent third party can make an independent verification.
In its present state the Etp report from the Hills group does a poor job of meeting this requirement.
Part of the process for developing a model is to subject it to tests to the natural world to see if it describes the real world adequately.
The Hills Group Etp model does not present any such tests.
Simple checks demonstrate that the Hills Group Etp model in its present form fails on this on all counts.
It is wished for that sites, like The Automatic Earth, SRSRocco and more, that has been peddling the Hills Etp report also posted this excellent analysis asap.
Hi Rune.
You said: “There is an unbroken rule within engineering and science;
A report should be structured and written in such a way that any competent third party can make an independent verification.
In its present state the Etp report from the Hills group does a poor job of meeting this requirement.”
How the hell would you know? You are not a competent third party. And you have no way of knowing whether SK is competent or even sincere. His “analysis” is of a straw man, not the Etp model. He might genuinely not understand the Etp model or he might have some other motivation to criticize it.
You also said: “Part of the process for developing a model is to subject it to tests to the natural world to see if it describes the real world adequately. The Hills Group Etp model does not present any such tests.”
Yes it does! The Etp model backcasts the average annual oil price from 1960-2013 with an accuracy of 96.5%. And the Etp model has correctly forecast the maximum affordable oil price and the downward price trajectory and rising inventories for the last 2 years and counting. In September of 2014, the Etp model forecast that today’s (2017) oil price would be $54/barrel. That prediction is exactly correct. So the Etp model does pass the test of describing the real world. Did you predict the oil price decline? Are you surprised by rising oil inventories?
Cunning, take your pills and pls give references to verifications of the Etp model from one or several independent third parties.
List oil fields/developments that now has EROEI (1.75?) as described by the Etp model.
Does the Etp model still predict $20 something oil by 2022?
Oh Cunning, read what shortonoil wrote back in February 2013;
“Following our graph, which we are 99% confident is accurate, oil prices by 2020 will be about $208/barrel, or $6.88/gal for transportation fuels. This is probably survivable, but by 2025 those prices will have advanced to $329/barrel and $10.90/gal. At some point the energy provided by a unit of fuel can no longer support the economic activity needed to acquire it. We have some graphs that address that issue, but in the final analysis it becomes an economist’s job to define it(we’re engineers).
Reviewing some of the other work that you have posted here, however, I would think that the impact would be significant (to the say the least).”
The Hill’s Group
http://www.theoildrum.com/node/9811/944630
Bullshit, Rune.
In February of 2013, the Etp model only had one curve: the Etp function. At that time, the Etp Maximum Oil Affordability curve had not yet been derived. In September of 2014, version 2 of the Etp model was published with both curves, and the model has been very accurate ever since.
Read what BW Hill was saying in that old quote again. He was not predicting that the oil price would keep rising. He was suggesting that it couldn’t. He was right. Once the Max Affordability curve had been derived from the Etp function, he no longer had to guess where the thermodynamic limit was.
Your post is totally disingenuous, another red herring. I notice you did not comment on or acknowledge your basic misunderstanding/misrepresentation of the Etp model that I just pointed out (down the page). Change the subject. Nice.
You also said: “List oil fields/developments that now has EROEI (1.75?) as described by the Etp model.”
That is silly. The Etp model does not suggest that individual oil fields have an ERoEI of 1.75. You just made that up. The Etp model is about the whole oil production system, not individual wells.
You also asked: “Does the Etp model still predict $20 something oil by 2022?”
I think the Etp model forecasts something more like a little over $11 for 2021. Do you find that hard to believe for some reason?
So why after all these years have not the Hills Group had its report verified by third parties?
“So why after all these years have not the Hills Group had its report verified by third parties?”
The Etp model is not groundbreaking new science. It is an engineering report based on basic thermodynamics. It’s premises and assumptions are not controversial. Just the results.
Conventional barrel counters like yourself are just surprised by how drastically the second law of thermodynamics affects oil depletion analysis. And I expect you are a little threatened by and perhaps jealous of the Etp model. After all, you’ve never made a forecast as accurate as the Etp model. And the oil industry certainly doesn’t want people to take the Etp model seriously. It is bad for business. How much do they pay you for disinformation?
So why do the results from the Etp model deviate so violently from real world observations and alternative approaches?
Rune, you said: “So why do the results from the Etp model deviate so violently from real world observations and alternative approaches?”
I have no idea what you are talking about. I already showed that the Etp model correctly forecasts the yearly average oil price limit and rising inventories. That does not deviate violently from real world observations. It is totally consistent with real world observations.
As far as alternate approaches go, they just don’t make real world observations that are anywhere near as accurate. As to the details of why that is, it might be fascinating to talk about, but I don’t see why such a question has any relevance to this discussion since we want to use the best method available. It’s like asking why we don’t use steam powered cars instead of gasoline powered cars. The answer is pretty obvious.
OK, perhaps you may help clarify
Report# HC3-433
Version 2, March 1 2015
“Depletion: A determination for the world’s petroleum reserve”
From The Hills Group
”2012 is the point where the petroleum production system (PPS) becomes a larger user of the energy that comes from crude oil than is the non-energy goods producing sector (NEGs) of the economy.”
Page 36
(I asked it before,but when the questions gets pointed you turn it into story time).
Also engineering reports are subject to third party verifications.
Since POB is fortunate enough to have such a rare expert on the Etp model as Cunning, perhaps he would be kind enough to describe what EROEI the quote from the Etp report below translates into?
Report# HC3-433
Version 2, March 1 2015
“Depletion: A determination for the world’s petroleum reserve”
From The Hills Group
”2012 is the point where the petroleum production system (PPS) becomes a larger user of the energy that comes from crude oil than is the non-energy goods producing sector (NEGs) of the economy.”
Page 36
Rune, you asked: “Since POB is fortunate enough to have such a rare expert on the Etp model as Cunning, perhaps he would be kind enough to describe what EROEI the quote from the Etp report below translates into?”
No. I have no real interest in this discussion.
And yes, you are very fortunate to have me comment at all here since I have been banned for life and the only reason I am even commenting now is because I just got a new IP blocker I am trying out. This oil industry funded disinformation site is not interested in having an honest discussion.
Could this be the one who called himself Futilitist and to my knowledge was banned at least 2 times at POB and also in several other places?
”And yes, you are very fortunate to have me comment at all here since I have been banned for life and the only reason I am even commenting now is because I just got a new IP blocker I am trying out. This oil industry funded disinformation site is not interested in having an honest discussion.”
That may explain the disruptive manners, lack of ability to look at other sources and personal attacks with every intention to obscure further informed debates.
Hi Rune,
He sure does sound like Futilitist, you are dead on about that.
Facts matter not at all to such a person.
This is just another witch hunt. Kill the messenger.
People should notice that I have made several corrections to the false arguments made against the Etp model. No one has answered or even acknowledged anything I said. They just change the subject and try another stupid, misleading attack.
People should also notice that no one here seems to understand that their arguments are ridiculous and childish. Dennis’ shameless ignorance of, and lies about, the second law of thermodynamics are stunning. Rune’s first comment about my post on entropy was to ask if I was Futilitist. Nothing on substance, just an attempt to scapegoat me. Now my old friend farmer mac joins in. What fun. This is obviously an oil industry shill site that denies the Etp model and soft pedals collapse.
http://futilitist.blogspot.com/2015/04/peakoilbarrel-soft-pedals-collapse.html
And it’s not the only one:
http://futilitist.blogspot.com/2016/12/peakoilcom-deception-etp-cover-up.html
And now we see a highly coordinated, multi-website hatchet job on the Etp model. Two fancy but highly fraudulent critiques suddenly appear, one in the US and one in Spain. And Ugo Bardi devotes his blog to promoting them and denouncing the Etp model. Amazing.
People are very afraid of the Etp model.
I left a comment for Ugo Bardi on his blog which is now awaiting moderator approval. I am printing it here just in case he chooses not to answer my questions:
“Dr. Bardi,
A couple questions:
If the Etp model is invalid, why did you recently allow Dr. Louis Arnoux to promote the Etp model on your blog with three rambling consecutive guest blog posts? It makes no sense. And have you looked into Dr. Arnoux’s background and credentials? He has been involved in some very shady, perhaps fraudulent business dealings which are very easily discovered with a simple google search. I look forward to reading your answers.
Thanks,
Futilitist”
I smell a rat.
Hi Futilitist,
No the argument in the post here shows that the ETP model implicitly assumes irreversibilities are either zero or must be equal to negative Q-dot divided by T at all times.
Many are mystified by Calculus, but not so much by algebra (I am not sure how good you are at Algebra, but I will assume a high school level of mathematics where most have mastered algebra).
For the first equation of the post I will change it to
a=b-c+d
the third equation is
b=-d, which is incorrectly stated as b=d in the Hill’s Group report (positive Q_dot means a flow of heat into the system in thermodynamics).
The only way for the first equation (based on the second law of thermodynamics and taken as given for the problem specified, no inlet and one exit) to be transformed to the third equation is to assume that at all times
a= -c
That can only be true if we assume d is zero, or equal to -b at all times.
In general, there is no physical reason to make this assumption.
This is the basic physical flaw of the ETP model.
Rhetorical devices and name calling convince nobody.
”This is just another witch hunt. Kill the messenger.”
Wrong! Do not play the victim card (It will not work!) when confronted with requests for documentation from the real world that validates the Etp model.
”People should notice that I have made several corrections to the false arguments made against the Etp model. No one has answered or even acknowledged anything I said. They just change the subject and try another stupid, misleading attack.”
Again wrong!
The thing is, anyone with some basic understanding of thermodynamics and the oil industry reaches independently of each other the same conclusion about the Etp model.
“It is nonsense!””
That is why the Etp model will get dismissed and it is the simple fact.
Cunning, so far you have failed at providing real world documentation that supports/validates the Etp model so you have none other to blame than yourself for not providing evidence instead of repeating some false mantra.
Rightfully you deserve all the flak that comes your way for promoting some false gospel.
Hi Futilitist,
Aside from where you have misinterpreted what I have said, can you give some quotes which substantiate your claims of:
Dennis’ shameless ignorance of, and lies about, the second law of thermodynamics
I am fairly well versed in thermodynamics having studied both classical thermodynamics (2 semesters in Mechanical Engineering) and statistical mechanics (1 semester as a Physics major), though not nearly to the level of Rune Likvern (and Fernando Leanme as well) who both dismiss the ETP model.
You fail to understand that in an open system the change in entropy can be negative while the surroundings of the system see an increase in entropy at least as large as the open system.
No physical laws are violated, it is simply a matter of where the system boundries are drawn.
Cunning wrote;
”How the hell would you know? You are not a competent third party. And you have no way of knowing whether SK is competent or even sincere. His “analysis” is of a straw man, not the Etp model. He might genuinely not understand the Etp model or he might have some other motivation to criticize it.”
http://peakoilbarrel.com/on-the-thermodynamic-model-of-oil-extraction-by-the-hills-group/#comment-596887
And now he claims being a “killed messenger”?
Cunning start providing evidence from the real world that confirms/supports the Etp model instead of attacking those who provides insights into it adequacy.
Rune, you demanded: “Cunning start providing evidence from the real world that confirms/supports the Etp model instead of attacking those who provides insights into it adequacy.”
One more time. I don’t work for the Hill’s Group. I am not their official representative. My opinions are my own. You are overtly hostile toward me and I don’t really feel like bothering with any of this crap. This place is a silo and an echo chamber. You guys are basically talking to yourselves. The answers you seek are readily available from the Etp model’s creator on the Etp Q&A thread at peakoil.com. Quit griping at me. I look forward to reading your upcoming dialog with BW Hill. Good luck.
Only reason I cared to participate on POB was that it presented a good post (this one).
Then Futilitist (after having being banned) unexpectedly showed up repeating his wild, still undocumeted claims about a report that by several others is thoroughly debunked. And more are to follow.
I got the report from a friend some months ago and after 30 minutes I found so many holes and inconsistencies in it that I was convinced it would die…and it has.
Your behavior made many think you worked for Hill.
Hey Rune Likvern.
Why are you still talking to me, Rune Likvern? Elvis has left the building. Please go to peakoil.com and sign up with your real name, Rune Likvern, so I can watch the fun. Are you afraid, Rune Likvern? LOL!
Or just make an excuse. Your call.
I got the 57% (per 2016) from someone following the Etp model.
50% is from the Hills group and was as per 2012.
http://peakoilbarrel.com/texas-update-november-2016/#comment-587645
”Falling production is likely to produce a cascading effect as 57% of the demand for petroleum now comes from the petroleum production system itself. Once production begins to fall, demand will fall, and accelerating declining prices will follow. This will push more producers past their minimum operating cash flow level. At that point a feedback loop of falling demand, driving falling prices, driving reduced production will take over.”
Hi Rune.
Those numbers are correct. Is there some reason to doubt them?
2012 was the energy halfway point, when it took the energy from 1 barrel of oil to produce 2 barrels of oil. Since then, the general economy has been starved of energy as production costs have continued to rise rapidly. The system is now in thermodynamic shut down mode and the price of oil is racing to zero as fast as it can. Civilization has begun to collapse, as I informed folks here back in 2015. That is why the world seems to be going mad and Donald Trump is President. This is what collapse looks like. So far. It will get a lot worse.
Yes, there is overwhelming empirical evidence to doubt the ETP. The oil industry from the well lease to the gasoline tank pathway is 80% not 43% energy efficient. The 20% loss is allocated as follows:
1. 8% at the well lease,
2. 1% transportation from the lease to the refinery,
3. 10% at the refinery,
4. 1% distribution from the refinery to the gasoline tank.
I think the Etp model forecasts something more like a little over $11 for 2021. Do you find that hard to believe for some reason?
I am not a fan of fossil fuels by any stretch of the imagination and I think that due to technological disruption there will be quite a bit of demand destruction which could certainly keep demand down and consequently prices low. However even I find a price of $11.00 a barrel by 2021 very difficult to believe.
Hi Fred.
You are way too in love with your technological disruption theory. You see it as the main cause of demand destruction. This is just wishful thinking. There is very little evidence to support such a position.
As far as the oil price being around $11/barrel in 2021, I found it difficult to believe at first, too. But, upon careful examination, the Etp model seems to explain the current situation better than any alternative theory. If I had told you in January of 2014 that the price of oil would be $54/barrel in February of 2017, you would not have believed that either. Yet here we are. The Etp model predicted it perfectly. And inventories are still rising, just as the model predicts they will. The world economy has contracted about 5.8% since July of 2014 when the oil price first collapsed. That is where real demand destruction is coming from. Oil can no longer pay for it’s own use in the economy due to it’s rising production costs. It is so obvious. Wake up, man!
”You are way too in love with your technological disruption theory. You see it as the main cause of demand destruction. This is just wishful thinking. There is very little evidence to support such a position.”
Over a hundred thousand Tesla cars already on the road, battery powered tools by the millions sold every week, wind and solar farms popping up like mushrooms in warm spring rains, every major car manufacturer gearing up to produce electric models on the grand scale, building codes being written already in some localities that mandate net zero energy houses, …………..
Cunning, thou art indeed a brazen liar, or else you’re just another idiot who doesn’t know doo doo from apple butter.
Here’s a link from WIRED which illustrates what the over the road truck of the future will look like, and it won’t cost more than maybe four or five percent more to build it than it would cost to build an equivalent sized new conventional truck.
BUT this experimental model gets over TWICE as many ton miles out of a gallon of fuel.
https://www.wired.com/2017/02/climb-inside-worlds-largest-wind-tunnel/
And when this sort of truck drives itself, as will happen within the next few years, and it can run slower than usual when traffic and schedule permit, it will do even better on fuel.
My last orchard sprayer had a two hundred horsepower engine, and she HOWLED like an old piston engine fighter, because than engine was driving a double thirty inch fan to create a powerful air blast that carried the pesticides from the nozzles all the way thru a row of trees on either side.
Now growers who are still working ( not retired) are switching to lower, smaller trees planted closer together, and they are using sprayers that need less than half as much power, and half as much water, to do the application job just as well, and just as quickly.
The amount of evidence indicating the possibility of substantial demand destruction is not only AMPLE, it’s OVERWHELMING.
Of course aggregate demand might still increase since the world population is still growing, and poor people are getting richer , and are able to afford more energy.
And since poor people who have never had electricity at all have never gotten used to having it twenty four seven three sixty five, they will be and are GLAD to start out with intermittent solar power, and work their way UP to reliable grid juice.
With a little luck, billions of very poor people will soon be able to afford a modest personal pv system and battery and a couple of lights, a radio, a super cheap computer for their kids, and some power tools that will greatly enhance their productivity, eventually a refrigerator, bigger tools, a tv, etc………….
I’m telling it like it is, and you either know it, or else………
Hi Futilitist,
It will be interesting to see what the oil price is in 2021.
I am fairly certain the average price of oil from Jan 1 , 2021 to Dec 31, 2021 will be above $12/b? So yes it is hard to believe barring a total economic collapse before that date.
Now when one assumes that this will occur, that oil price of $11/b (in 2021 $ as the Hill’s group does not believe in adjusting for inflation) might sound reasonable. Under the more likely scenario that the World economy will continue to grow at about 4% per year (in nominal terms rather than real terms), it is very unlikely that if oil supply grows at less than 1% per year, that oil prices will remain low. My expectation is that oil supply will be short if oil prices remain low (under $50/b) and that generally a shortage of a good tends to result in increased prices.
I realize that you think thermodynamics determines prices, and have faith that the Hill’s group understands thermodynamics better than the professors who taught them, but time will show you are incorrect on both counts.
Dennis,
You said: “I am fairly certain the average price of oil from Jan 1 , 2021 to Dec 31, 2021 will be above $12/b? So yes it is hard to believe barring a total economic collapse before that date.”
What a stupid word game! You are “fairly certain” but you don’t actually explain why. Because you can’t. It is just your completely unjustified opinion. Then you try to hedge by saying it is hard to believe “barring a total economic collapse”.
Well, I have some really bad news for you, Dennis. We are in the early stages of a total economic collapse. This began at the end of June of 2014, when the oil price collapsed. Oil no longer supplies enough net energy to pay for it’s continuing production. Inventories keep growing and the oil price keeps falling exactly as the Etp model has been predicting for the last 2.5 years running! You have been either uselessly vague or completely WRONG in every forecast you have made during the same period. Why should anyone believe you?
You also claimed: “Under the more likely scenario that the World economy will continue to grow at about 4% per year…”
What the hell are you smoking? World GDP has fallen by about 5.8% since the oil price collapsed:
http://data.worldbank.org/indicator/NY.GDP.MKTP.CD
You can’t just dismiss the possibility of $11/barrel oil in 2021 (for no particular reason at all), and then blithely turn around and suggest that the world economy will continue to grow at a 4% rate (for no particular reason at all) when it is actually currently shrinking. You are just dissembling and trying to spin false arguments. You have an agenda. It is disinformation and it is really obvious. Your paymasters would be wise to find a more competent liar.
World GDP dropped 1.7% in 2009, during the deepest recession since WWII. This was the only post-war year when global economy was declining. Since then, global GDP was growing. In 2015, growth was 2.63% (at market exchange rates).
http://data.worldbank.org/indicator/NY.GDP.MKTP.KD.ZG
PPP-based world GDP growth was 3.2% in 2015 and 3.1% in 2016. IMF expects it to accelerate to 3.4% this year, and 3.6% in 2018.
http://www.imf.org/external/pubs/ft/weo/2017/update/01/
$12/barrel oil implies a collapse of global civilization.
At this price, only about 10-15 mb/d can be produced profitably (vs. current consumption of 97 mb/d).
If you look at your chart, it says -1.7%.
That’s minus 1.7%.
That’s a greater drop from the previous year than ‘1.7%’.
Looking at that graph, there appears, nonetheless, to be a downward trend.
If civilization is collapsing fractally, which it likely is, then ~$12 a barrel at some point or points, wouldn’t seem too far-fetched, such as if one of the next dips sees > -4%.
Caelan,
I said “World GDP dropped 1.7% in 2009”
Isn’t it clear that this means -1.7% ?
But that was the only post-WWII year when global GDP declined.
Global GDP growth (%)
source: World Bank
Thanks AlexS
The nominal World GDP dropped in 2015 (in current US $) due to World deflation, based on World Bank data.
If we take the data for World nominal GDP in US $ and take the natural log to find average growth rate for the past 25 years, the average rate of growth has been 5.33 % per year for nominal World GDP in current US $. For real GDP (constant US $) it has been roughly 3% average growth.
Dennis,
Who cares about nominal global GDP in US$?
What’s important is real GDP growth
AlexS,
Only it dropped even more than -1.7, since we are talking about a previous positive number.
That’s quite a drop and looks like it’s going to give a good downward pull on the trend.
In any case, that you suggest that it was the only post-WWII year when global GDP declined seems kind of ominous. I guess we should expect to see increasing occurrences of the minus sign.
I should have phrased the question if global EROEI now was about 1.75 as referred by some who follows it closer than myself.
I also gave reference (further down) to where I had the number from.
In other words I did NOT make it up.
Perhaps you should spend some time on learning manners instead of making false accusations.
And you still have not come up with any real life references that validates the Etp report, after all it is science/engineering so that should be quite easy.
It does not really matter much for the conclusion if the global EROEI for petroleum systems derived from the Etp model now is 2 or 1.75.
No reason for you to throw a temper tantrum and come with insults.
I stopped trying to make sense of the Etp model when I was insulted by “Shortonoil” on the Peakoilnews website for asking a relatively simple question. The behavior of “Cunning/futilitist” on this forum confirms to me that there is more ego than science at the Hills group.
JJHMAN,
I do not work for the Hill’s Group. I just genuinely believe that the model is valid.
Beyond that, I am very fascinated by the coordinated effort to discredit the Etp model. For the last 2 years, I have heard every argument against the Etp model. They read like an ignorant, rambling, unphysical, and largely self contradictory laundry list of irrelevancies. Now, all of a sudden, two authoritative looking negative reviews appear simultaneously in the US and in Spain. Both reviews are just the same stupid laundry list of lame objections dressed up in fancy sounding physics language. At the very same time, Ugo Bardi publishes a cheesy hatchet job on the Etp model that promotes those two bogus papers, after previously offering his blog as platform to help Dr. Louis Arnoux promote the model.
When Ugo Bardi first published Dr. Arnoux’s essays, I thought it was a set up. It appears I was right about that. Here is my conversation about all this so far with Ugo Bardi from his blog’s comment section:
FUTILITIST February 26, 2017 at 8:37 PM
Dr. Bardi,
A couple questions:
If the Etp model is invalid, why did you recently allow Dr. Louis Arnoux to promote the Etp model on your blog with three rambling consecutive guest blog posts? It makes no sense. And have you looked into Dr. Arnoux’s background and credentials? He has been involved in some very shady, perhaps fraudulent business dealings which are very easily discovered with a simple google search. I look forward to reading your answers.
Thanks,
Futilitist
Ugo Bardi February 26, 2017 at 11:41 PM
As I say in my post, Arnoux’s texts are qualitatively correct and they do identify a fundamental factor in the decline of the system.
FUTILITIST February 27, 2017 at 12:13 AM
But you also say that catastrophism is bad. Dr. Arnoux’s conclusions are clearly catastrophic. Yet you published his 3 guest posts and you now endorse his position as “qualitatively correct”. You now claim to just have a problem with the Hill’s Group report that it is based on. Can you see why this might seem a little inconsistent?
How did you come to publish Dr. Arnoux’s posts in the first place?
Ugo Bardi February 27, 2017 at 3:00 AM
Why are you so angry? I just thought that Arnoux’s posts were interesting for the discussion and they did highlight a fundamental point. Catastrophism is bad when it is based on faulty data and interpretations, but we do face catastrophes so my point is that we should call wolf only when we have good data showing that the wolf is really coming. Then, we all try to understand what’s going on, out there, and none of us is infallible.
I see that you accuse Arnoux of shady and fraudulent business dealings, that may be, but please note that, 1) I know nothing about that and 2) Arnoux didn’t use his posts to promote business deals (apart from the $28 cost of the report!)
FUTILITIST February 27, 2017 at 9:24 AM
Why would you say that I am angry? I just asked you a couple of simple questions.
I asked you how you came to host Dr. Arnoux’s 3 guest blog posts in the first place. If you are unfamiliar with Dr. Arnoux’s background, how did his guest posts get published on your blog? Is he your friend? Did he contact you? Did you contact him?
Did you believe Dr. Arnoux was accurate at the time you published his 3 posts? If not, why did you publish them? If you did believe that Dr. Arnoux was accurate, but now you don’t think so, what caused you to suddenly change your mind?
Ugo Bardi February 27, 2017 at 9:30 AM
Come on, Futilist, how many times are you asking me the same questions? And totally irrelevant questions: whether Arnoux contacted me or I contacted him, or if he is my friend. What does it matter? Calm down, please.
FUTILITIST:
With all due respect, Dr. Bardi, you are dodging my questions. You just said that you published Dr. Arnoux’s posts because they were interesting to the discussion. Yet, to my knowledge, you are the first to publish Dr. Arnoux’s posts. If his posts have not appeared anywhere before you published them, how did you become aware of Dr Arnoux and decide to publish his posts?
——–
My last question is still in moderation. I wonder if Ugo Bardi will ever answer it.
Conspirare — to breathe together.
BTW—This is the second time I am posting this because the first time I posted it, you flagged it as spam. This censorship! If my second try fails to appear, I will publish it on my blog. Thanks.
Loren,
Many of us, if not all, sometimes get odd stuff happening with how this site treats our comments. The past week has been especially odd in that regard, so it’s not exclusive to your comments.
In any case, it’s The Hill’s Group and/or Bedford Hill’s model, not yours. So shouldn’t he/they be defending, at least more than you, their own model?
As mentioned in one of my comments, money/price is symbolic. It therefore has a disconnect from reality– by definition– and therefore a disconnect from thermodynamic reality.
That said, humans can exhibit serious disconnects from reality, too, such as through their symbolic forms, and sometimes they may not even be aware of it.
Conspirare — to breathe together.
Nice edit, BTW.
Conspirare sounds nice, agreed and thanks.
Cool it, Rune.
You said: “In other words I did NOT make it up.”
I never said you made it up. I said: “Those numbers are correct. Is there some reason to doubt them?” That means I am confirming and agreeing that your numbers are correct (to the best of my knowledge). I am just asking why you think those numbers are wrong, besides the fact that you personally find them astonishing.
I am not being rude in any way. Accusing me of rudeness is just a game.
Will you now confirm that global (weighted) EROEI now is 1.75?
You are aware that since there must be a spread on individual fields around the weighted average, there is nothing silly about asking for documentation/references to real world fields that have low EROEI?
Your primary objective on POB has been to try to derail, insult and harass anyone providing evidence that pokes holes in the Etp model.
“I am just asking why you think those numbers are wrong,”
Because every other credible sources, governments, oil companies, academia, observations of the real world proves an EROEI now of 1.75 (or 2 and at the wellhead) has to be wrong.
And you after having studied the Etp model for more than a year have not provided any documentation that proves ALL the other sources wrong.
Rune, you said: “You are aware that since there must be a spread on individual fields around the weighted average, there is nothing silly about asking for documentation/references to real world fields that have low EROEI?”
The Etp model includes refining and transportation, not just the ERoEI at the well head. So you are asking an invalid question. Do you understand?
Question still relevant as there is nothing from other authoritative sources that now puts average EROEI at 2 from reservoir to consumer.
As a matter of fact with an global weighed EROEI of 2 (from reservoir to consumer) all evidence suggest there would be no/little oil production.
I presented a case with Volve further down which was shut down with EROEI of 7-8 from reservoir to consumer.
Rune,
I don’t work for BW Hill or the Hill’s Group. I am expressing my own understanding of and opinions about the Etp model. I explained before that I am not really interested in having this detailed debate with you. I don’t know what your latest point is. It would require some work on my part to understand what your complaint on this particular point actually is. But you have shown a strong tendency to misunderstand/misrepresent what the Etp model claims or how it works, so I feel very hesitant to take this discussion seriously. I am sorry you are so upset about this.
If you really want to have an official discussion about all the details of the Etp model, I would recommend that you go to peakoil.com and talk with the model’s creator, BW Hill. He is the one who can give you the definitive answers you seek.
On Topic: JB covers it all, Oily Reality even on Conservative Radio.
http://www.stitcher.com/podcast/john-batchelor/the-john-batchelor-show/e/the-race-for-whats-left-the-global-scramble-for-the-worlds-49181295
I have asked Dennis Coyle to correct a couple of errors. Namely, the second equality sign in the first equation ought to be replaced by a plus sign. The second equation as well as the third pair of equations should have minus sign after the equality sign. Also, clearly “If one the other hand” ought to be “If on the other hand”.
Thanks for all the positive comments. Rune especially makes the point similar to that attributed to Bacon; namely that truth emerges better from an outright error than from confusion. The confusion in the Hills Group report makes it harder to discover the fundamental errors, particularly since not many are familiar with exergy analysis. As I mentioned in my summary, Michael Moran is the one person who systematized the exergy analysis and included a chapter in his thermodynamics book. He wrote an earlier book entitled Availablity Analysis, a Guide to Efficient Energy Use, in 1982. As a result, those undergraduate thermodynamics courses that include the chapter in exergy analysis equip the students to use the exergy balance equation with the same facility they use the first law of thermodynamics. But pressure to reduce the number of courses in thermodynamics in mechanical engineering education has meant that today there is only a single semester course (as opposed to two 50 years ago). Hence the exergy analysis today tends to be dropped from the syllabus. In any case, the subject matter is undergraduate level material and now readily accessible in the excellent textbook by Moran et al. If it turns out to be more powerful than what is thought today, it will certainly find more use in the future.
One easy way to illustrate energy flows is by the use of Sankey diagrams, which illustrates were energy goes from source to end user.
Simplistic explained the Hills report does not include the chemical energy embedded in oil as it is extracted from its reservoir. The use of temperatures with depths is a red herring.
There is potential energy (pressure) in the reservoir that helps drive the oil/gas to the surface and then there is heat from the surroundings embedded in the fluids extracted from the reservoir, both oil, water and gas (if any).
With a few and very rare exceptions, this physical energy (heat and pressure) from the reservoir does not make it to the end consumer.
In some rare cases this physical reservoir energy may be used at or by near by facilities as the extracted reservoir fluids are cooled down (heat removed) during the stabilization and separation of the products. Natural gas through a turboexpander.
Oil is cooled down before storage or transport.
Just a question or three.
How much oil would we get outof the ground without it being under geological pressure and / or hot? OK, technology inputs (water or gas or fracking) keep the stuff coming. At some point the drillers give up or go somewhere else? We need a calculation of the cost curve(s) for this procedure. I presume at some point the energy expenditure – drawn partly from the background industrial economy, rises above the economic limit. Presumably this correlates critically with net chemical energy in the oil we are still getting?
What is your answer to your own question? (“How many percent would net energy drop as EROEI declines from 15 to 10?”) My guess is one third. Does that mean one third more drilling is needed to energetically fund the same external economic activity?
Without primarily the over pressure it would become much harder to get the oil/gas out of the ground.
The limits the economies will encounter is the replacement costs for oil and gas and at some point availability.
Costlier oil/gas may be seen as a proxy for lower EROEI.
With an EROEI of 15 a surplus of 14 units is available after 1 spent, stated otherwise 93.3% is available from what was totally recovered.
With an EROEI of 10 a surplus of 9 units is available after 1 spent, stated otherwise 90.0% is available from what was totally recovered.
In other words, the drop in available energy with EROEI dropping from 15 to 10 is:
3.3/93.3 * 100% = 3.5%.
The above is one reason EROEI gets little attention as it takes small increases in gross energy output to sustain or grow net energy.
And when it comes to drilling it is about the size of recoverable reserves in the discovery.
Thank you.
It has surprised me reading different interpretations of EROEI over the years. I agree that small increases in gross energy output are required to sustain the total supply of net energy (in this case fungible oil within a global industrial economy).
But I wonder how this looks to you? Suppose the global industrial economy, arguably, ‘needs’, in total, 60 units of continuous supply. At 15:1 the extraction industry needs 4 of those units. If EROEI drops to 10:1 but the global ‘need’ for oil stays at 60 units, the extraction industry must now take 6 of those units. Thus the extraction industry’s energy costs are raised by 50%.
In your case available energy drops from 56 to 54, that is a drop of 3.5%..
Rune, you said: “Simplistic explained the Hills report does not include the chemical energy embedded in oil as it is extracted from its reservoir. The use of temperatures with depths is a red herring.”
Once again, if the mass (oil and water) and heat removed from a control volume (oil reservoir) are known, then the Entropy Rate Balance Equation For Control Volumes can be used to calculate the exact amount of energy expended in the process. Simple. This is the Energy Invested (EI) part of the ERoEI of oil.
The chemical energy of the oil, released through it’s combustion, is the Energy Returned (ER).
So, when you say: “With a few and very rare exceptions, this physical energy (heat and pressure) from the reservoir does not make it to the end consumer”, you are the one arguing a red herring. The Etp model in no way suggests that heat and pressure ever make it to the end consumer. That has nothing to do with the model or the actual claims of the model. It is just silly and irrelevant. Please review this again to see why you are not actually critiquing the Etp model, though you seem to think you are.
Cunning, why not make it easy on yourself and point us to where in the report we can find the things we question?
A couple of more things
Report# HC3-433
Version 2, March 1 2015
“Depletion: A determination for the world’s petroleum reserve”
From The Hills Group
” The extraction, processing and distribution energy costs for 2012, when summed, equal 70,000 BTU/gal; which is what the ETP model predicts.”
Why does this not show up in the financial statements from the oil companies?
” It therefore follows that average extraction costs are close to sale price.”
Page 49
And where in the report are the equations that describes input from other energy sources like natural gas, coal, nuclear, hydro other renewables?
what would the oil companies financial statements look like if they would be increasing their reserves instead of pumping them dry?
The Saudis wanting to ipo Aramco smells to me like desperation. Could it be an attempt at using funny Money to prop up the disaster for just a bit longer?
Bingo.
Try to behave like an adult.
Um, Rune, stop playing games. I just agreed with farmboy. Is that okay with you?
I posted a relevant question and so far no one have tried to answer it.
But that is and have been your MO when real world observations runs contrary to your belief.
Rune,
I have tried to patiently answer all of your questions. Stop attacking me personally, please.
farmboy,
And how is that related to my post?
Oh, with financial statements I also meant P&L statements.
Now see if you can formulate an answer to my post.
Rune your mental capacity and education related to the oil industry is well beyond mine, it just seems to me you have an agenda.
Thankfully I have no need to formulate an answer to your post since thats beyond my abilities. Fortunately Shallow Sand, Ron, Mike, Petro…. have posted plenty in the last couple of years showing the awfull numbers behind the glowing reports coming out of significant portions of the oil industry.
How can it be that The Hills Group correctly forcasted the drop in crude prices back in sept 2014 when the price was still above $90 wti? their forcast is still holding with uncanny precision 2 1/2 years later. Why didn’t you inform us of the halving in crude prices?
You’re not suggesting that the ave oil production was making a profit of more than $45/barrel when prices were still above $90?
”Fortunately Shallow Sand, Ron, Mike, Petro…. have posted plenty in the last couple of years showing the awfull numbers behind the glowing reports coming out of significant portions of the oil industry.”
Yes, they have done a great job at that.
The question is about the cause for the poor numbers. The poor numbers have been for companies engaged in costlier oil (a proxy for lower EROEI oil). The investments in costlier oil is not profitable at lower oil prices, thus the poor results.
However, looking at specific operational expenses (LOE) for shale companies these are on par with majors (primarily in conventional oil).
LOE includes costs for energy (primarily electricity) used in oil field operations and if the case was that the EROEI for petroleum systems now is about 2.0 from the reservoir to consumers, that is, for about 2 barrels of oil produced, the energy equivalent to 1 barrel of oil is used in the value chain from reservoir to consumers, split at about 0.7 barrels at the wellhead and 0.3 barrels for transport, refining and distribution.
0.7 barrels of oil equate to about 420 – 450 kWh electricity at the gate and for US industrial users it costs (EIA) $0.069/kWh, thus about $30 in electricity costs or about $15/bo produced.
LOE costs, which include energy, maintenance, water treatment and disposal, gas conditioning, etc. is by the companies reported in the region of $4 – $10/bo.
This documents that the claim about EROEI now at 2 is flawed.
In short, my point is that if the EROEI is as low as 2, this would also show up in the Profit & Loss statements of the companies. It does not, which drives another big hole in the ridiculous estimates from the Etp model.
If EROEI was 2 then the US crude oil producers and refineries together would use more electricity than EIA reports for the US Industrial Sector.
They are not!
Then add in NGL and natural gas production.
Perhaps some of the stripper well operators (low EROEI producers) have data on how much energy/electricity they use for each barrel of oil produced.
”How can it be that The Hills Group correctly forcasted the drop in crude prices back in sept 2014 when the price was still above $90 wti? their forcast is still holding with uncanny precision 2 1/2 years later. Why didn’t you inform us of the halving in crude prices?”
Luck/accident! And changing the narrative as time passes.
Did they predict the $60/b in May-15 and the below $30/b in Feb-16?
There were lots of analysts (and oil companies) that saw that one coming in early 2014 (using different metrics than thermodynamics) and the smart ones did not publish it.
It is not possible to project the oil price into the future based on a flawed thermodynamic model.
Back in 2012 I (in public) predicted the oil price would come down (but impossible to say by how much and exactly when), the point is that the multitude of parameters that participates in the price formation for oil are impossible to model except if the good ones are chosen oil price trends and approximate levels are possible to make reasonable projections of.
The demand is one of the tough ones and demand relates much to monetary and financial policies (these are two of the important ones), perceptions of the future, total debt levels, interest rates, exchange movements (above ground factors) etc. which again illustrates the irrelevance of a thermodynamic model to predict future oil prices.
Going forward I expect the oil price to remain low ($50-$60) and it could temporarily go lower and at some point the market will rebalance that will support a higher oil price (fundamentals always work in the long run), but to predict exactly when is difficult, it could take 1 year, 2 years.
As investments have been and now are low, global oil production will decline at some point in time and to offset this the price have to come up to make the companies invest more.
Oil will be the last industry to fail.
Your last question?
farmboy skeptically asked: “Rune your mental capacity and education related to the oil industry is well beyond mine, it just seems to me you have an agenda…Why didn’t you inform us of the halving in crude prices?”
Rune Likvern freely confessed: “There were lots of analysts (and oil companies) that saw that one coming in early 2014 (using different metrics than thermodynamics) and the smart ones did not publish it.”
Shocking. The oil industry analyst, Rune Likvern, just casually informed us that our worst suspicions about oil industry analysts are all true! Oil industry analysts have an agenda and we cannot trust them to tell us the truth (even if they actually knew what the truth was, which they clearly don’t).
You are correct “The demand is one of the tough ones”. The ETP model correctly predicted that demand will decrease.
“The Maximum Affordability curve, which is derived from the Total Production Energy function of the Etp Model, calculates what the maximum sustainable price of WTI for the following years will be:
2o15…$77
2016…$66
2017….54
2018….41
2019….27
So they post a price ceiling but no floor.
Your misrepresentation of the ETP by posting “In short, my point is that if the EROEI is as low as 2, this would also show up in the Profit & Loss statements of the companies. It does not, which drives another big hole in the ridiculous estimates from the Etp model.” can only be due to complete ignorance or trying to deceive.
If you don’t even understand the difference in EROEI and the ETP than why should anybody trust your evaluation of the thermodynamics?
My understanding is that an EROEI of 7:1 is closer to the current situation of an ave well.
“The ETP model correctly predicted that demand will decrease.”
Last year, global demand increased by 1.6 mb/d. This year, it is expected to increase by 1.4 mb/d. Both numbers are above 15 years-average.
Where is a decrease in demand?
Farmboy, further up in this thread I posted and I will repeat it here for you:
The Etp report says that
Report# HC3-433
Version 2, March 1 2015
“Depletion: A determination for the world’s petroleum reserve”
From The Hills Group
”2012 is the point where the petroleum production system (PPS) becomes a larger user of the energy that comes from crude oil than is the non-energy goods producing sector (NEGs) of the economy.”
Page 36
Note this is a result from the model presented in the report, not a claim or a working theory put to test.
” The extraction, processing and distribution energy costs for 2012, when summed, equal 70,000 BTU/gal; which is what the ETP model predicts.”
” It therefore follows that average extraction costs are close to sale price.”
Page 49
What EROEI at the consumer level do you estimate the conclusions from the Hill’s report comes up with?
Where in the report (page) does it say EROEI is 7? And at where?
Giving a list of ceilings to the price does not prove the validity of the report.
What would the consequences be of a max oil price of $27/b in 2019?
And if you believe the Hill’s report, go and short oil.
No one is interested in your understanding, but what evidence you produce.
Rune, please stop being so deceptive or you will Rune your reputation even more than you have already!
You asked: “What EROEI at the consumer level do you estimate the conclusions from the Hill’s report comes up with?”
We have been through this over and over. You yourself calculated from the 57% number that it would yield an ERoEI for oil of 1.75 to 1 (at the consumer level). I agreed that your number sounds about correct (you should ask BW Hill to confirm it). Why the hell are asking this question again? Why don’t you believe that this number is correct?
You also asked: “Where in the report (page) does it say EROEI is 7? And at where?”
Why the hell are you asking this again? It does not say that the ERoEI is 7 (at the well head) anywhere in the report. It has been explained to you more than once that the Etp model does not give such a breakdown. The Etp model is based on the Entropy Rate Balance Equation for Control Volumes which, in this case, yields the TOTAL energy used to produce, refine, and distribute oil and it’s products. I believe BW Hill has, separate from the Etp report, calculated the number you keep asking for. I don’t personally know exactly how he has done this. If you want to know how he arrived at that number, you will have to ask him yourself.
You misleadingly said: “Giving a list of ceilings to the price does not prove the validity of the report.”
No one claimed that it does. You tried to claim that the model did not correctly predict the price bottom of $26/barrel. Farmboy corrected your false and misleading claim. The price ceiling that farmboy just showed you is the Etp model’s official MAXIMUM price forecast. Do you know what MAXIMUM means? The model does not predict the exact price, just the yearly average MAXIMUM price. The model has been completely accurate in this forecast for the last 2.5 years running. The last time I checked, $26 is below $54. You are just trying to confuse the readers. Once again, the Etp model does not claim to forecast the exact oil price, just the MAXIMUM oil price. The average annual oil price has been well below the Etp maximum price curve for the last 2.5 years. Thus, the forecast is 100% correct. Do you understand?
For some weird reason you asked: “What would the consequences be of a max oil price of $27/b in 2019?”
Use your imagination. It will certainly be very bad for the oil industry and for civilization as a whole. It might trigger the rapid collapse of modern industrial civilization before we even get to 2019.
You also petulantly retorted: “And if you believe the Hill’s report, go and short oil.”
That is what I am doing.
And finally you postured: “No one is interested in your understanding, but what evidence you produce.”
And no one is interested in your deceptive bullshit anymore, Rune Likvern.
“the awfull numbers behind the glowing reports coming out of significant portions of the oil industry.”
This “significant portion’ (tight oil) accounts for only 5-6% of global oil production. And their awful results were due to unsustainably high growth rates, resulting in capex significantly exceeding operating cashflows and rapid accumulation of debt. With growth rates at 2-5% per year even shale companies could be net cash generative and profitable.
Rystad examines what to expect from U.S. shale break-even prices in 2017 this may surprise of few of you?
http://www.worldoil.com/news/2017/2/28/rystad-examines-what-to-expect-from-us-shale-break-even-prices-in-2017
money shot: “This means that for the first time the investments did not exceed the cash from operations for the shale companies. The shale companies have been able to reduce the imbalance between cash from operations and investment from $16 billion in first-quarter 2015 to zero in third-quarter 2016. This has been achieved by a considerable reduction in investments. For 2017, Rystad Energy forecasts an average WTI oil price of $60/bbl, which implies a 40% improvement in the cash from operations. This improvement in the cash flow will result in higher investments of the shale operators.”
The fact that many shale companies have reached cash neutrality in 2H16 ( and some of them were even slightly net cash positive) does not surprise me, as I have seen quarterly results of several LTO producers.
Cash neutrality was achived due not only to lower costs, but also to much lower capex. And lower capex resulted in lower production volumes.
In 2017, shale companies are planning to increase capex by 25-30%. And projections from many sources (including Rystad, IHS and others) suggest a return to service cost inflation. So cash neutrality threshold for shale producers will be much higher this year vs. 2H2016.
Rune, you asked: “Cunning, why not make it easy on yourself and point us to where in the report we can find the things we question?”
Simple. Because you keep asking for things that are not in the report.
You also asked: “Why does this not show up in the financial statements from the oil companies?”
Are you serious? Because financial statements are largely bullshit and physics is science.
And you also asked: “And where in the report are the equations that describes input from other energy sources like natural gas, coal, nuclear, hydro other renewables?”
I find this question particularly frustrating. It is simply not a valid question, and it just shows that you still do not understand the model you are attempting to criticize. I have repeatedly explained that the Entropy Rate Balance Equation for Control Volumes outputs the TOTAL amount of energy lost to the system (entropy) when a known amount of mass and heat are removed from the control volume. It does not give a breakdown of where the energy comes from. Just the total energy needed to lift the oil. So there are no equations that describe inputs from “other” energy sources, because such equations are not necessary. All sources of energy are already accounted for in the TOTAL energy used. And deriving the TOTAL energy used to produce, refine, and distribute oil is the whole point of the Etp model. Do you understand?
Hi Futilitist,
What we understand is that the ETP model makes assumptions which are not justified (and not likely to be correct). The results from such a model don’t match the data.
Now one can propose that when the observations don’t match the theory, that the observations must be wrong.
Science generally makes careful observations and requires the theory to match the observations, when this is not the case the theory is discarded and we work on a better theory.
SK has actually tried to engage Hill to improve the theory, but now believes the theory is not workable.
Yes the oil industry definitely funds all peak oil sites so that industry funding dries up. 🙂
”Are you serious? Because financial statements are largely bullshit and physics is science.”
That does not qualify for a serious answer.
Try again!
The thing is, each time the Etp model has an encounter with reality it breaks down. And the ones clinging to the Etp models gospel goes in full shut down.
“It does not give a breakdown of where the energy comes from. Just the total energy needed to lift the oil. So there are no equations that describe inputs from “other” energy sources, because such equations are not necessary.”
That is one of the big flaws of the Etp model.
”Just the total energy needed to lift the oil.”
So how big portion of the total energy used/gained in the process by said model is used for lifting the oil?
That should be in the Hill report.
The Entropy Rate Balance Equation for Control Volumes gives the TOTAL energy used only. Do you understand? It does not give a breakdown of where the energy comes from. I have stated this over and over, yet you persist in asking for such a breakdown. Why?
The Etp model has been making an accurate forecast of the continuing oil price decline in the real world for more than 2 years running. You haven’t ever made a prediction that accurate in your entire life! Yet you are sure the Etp model must be wrong, even if you don’t understand how it works. Lame.
Why don’t you take all the factors that you claim are missing from the Etp model report and construct your own model that forecasts the oil price better than the Etp model. If you can do that, I will take your claims very seriously. Until then…
Cunning wrote;
”Once again, if the mass (oil and water) and heat removed from a control volume (oil reservoir) are known, then the Entropy Rate Balance Equation For Control Volumes can be used to calculate the exact amount of energy expended in the process. Simple. This is the Energy Invested (EI) part of the ERoEI of oil.
The chemical energy of the oil, released through it’s combustion, is the Energy Returned (ER).”
Can you elaborate a little further on what the EI describes?
Is that the energy required to get one barrel of oil from the reservoir to the wellhead (or ex the installation)?
Does this include energy expended from support facilities?
It includes all the energy used to produce, refine, and distribute oil and it’s products.
I have said this many times before.
If, after 2 years, you still don’t understand this basic fact about how the Etp model is constructed, how can you be so sure the model is invalid? Your argument is a straw man if you keep misrepresenting what the model actually says.
Good with clarifications, but it does not change the fact that the Etp model is hugely flawed.
And you are fully entitled to your opinion even if you cannot actually justify it. I really don’t care.
You have wasted the last 2 years complaining about a model you don’t even understand. If you are sincerely interested in evaluating the Etp model, please take my advice and ask BW Hill your questions directly. He can be found at peakoil.com on the Etp model Q&A thread.
CL,
Your patience and persistence are admirable, but you are wasting your effort. As much as I admire Rune and Dennis, I’m not sure they will ever understand the concept. Cut them some slack as most people seem to get fixated on the trees instead of the entire forest ecosystem with total energy flows.
Having been educated in systems ecology, I don’t get hung up on the numbers and calculations, as they are simply tools for evaluating the processes and energy flows. It is the trends, critical linkages, and tipping points that point to potential outcomes that are significant.
The ETP model surely isn’t perfect, but it gives a big picture view I’ve not seen elsewhere, and it makes sense to me without quibbling about numbers. Time will tell.
Gotta agree on one thing. When there is this much concerted effort to discredit something, it smells bad.
Don’t know much about the folks at Hills Group, but they must have very thick skins. It is tough when you know you are right, but the world doesn’t want to understand, and would like to harm the messenger.
Please be nice so they don’t kick you off the site again. You’ll catch more flies with honey than vinegar, and I value your comments.
For starters, it probably isn’t a good idea to accuse the blog of being a shill for the oil industry. A few commenters aren’t all of us, and everyone you wrongly paint when you stereotype dislikes it. OK to think it, just please don’t write it and alienate everyone.
Wait and see, the ETP Model will evolve, improve, and may prove to have been a useful tool in hindsight, or not. Either way, few people will understand the systems concept.
Jim
Hi Jim.
Thanks for the kind words. It is good to hear from a real person like yourself.
As far as playing nice and not mentioning the obvious elephant in the room (i.e. that this is an oil industry shill site), I don’t think I can do that. I feel that I am doing a public service here. Sites like this exist to produce propaganda. They feed off of the comments of real people who are largely unaware of this or don’t have the level of commitment that the trolls and shills do. People who actually attempt to highlight and oppose this ongoing scam are summarily banned, like I was. The net result of all of this is a steady stream of fake news.
Real people who come here should take no personal offense when I point out that this site is a scam. Real people should feel no tribal connection to this sad place. This is not a real “community”. Superficial rules of politeness and decorum are just being hypocritically used against us to make it harder to find the truth. Resist.
— Loren
Hi Loren,
I noticed after accusing me of lying etc, you produced no evidence (direct quotes not taken out of context) of this.
I also asked you to share your physics knowledge to analyze the entropy change of a hot object (100 C) as it cools to the same temperature as its surroundings (20 C). There is no flow of material, just a flow of heat in this scenario.
The “system” in this case is the hot object.
Can you tell us what the change in entropy of this system is? Is it positive negative or zero. Assume the temperature change is 80 C, T0=100 C, and Te=20 C.
Thanks.
Hi Cunning Linguist,
So by your lack of answer, I will assume that you concede that for non-isolated systems it is entirely possible that entropy can decrease.
The claim that all natural systems will always increase in entropy is false, it depends upon the boundries around the system and whether the system is isolated.
You aren’t trying to claim that the Petroleum Production System is an isolated system I hope as that is far from being the case.
Note that I have sought answers from Hill at Peak Oil, but he doesn’t really have a very good grasp of thermodynamics.
There is no such thing as negative entropy, Dennis. As the hot object cools, it’s ability to do work decreases, meaning it’s entropy rises. Do you understand? Entropy is always positive or zero (in idealized machines). Entropy can be exported, as in living systems, and this is sometimes referred to as “negative entropy”, but it is not actually negative entropy. There is no such thing as negative entropy.
Oil production is a PROCESS. All processes produce entropy. Entropy rises. This is the second law of thermodynamics. The point is that irreversibilities (entropy) build in the oil production process. Your distinction between isolated and non isolated systems is a red herring. The oil production process is not a reversible process, is it?
As far as BWHill or myself not wanting to bother with your ridiculous claims about negative entropy, it certainly doesn’t call into question anyone’s lack of physics knowledge but yourself! Why should any serious person waste their time dealing with your relentlessly inept disinformation campaign?
By the way, Physicsnerd at peakoil.com already did debunk your false claims, but you failed to answer him there and you failed to mention his response here. This is very misleading and decptive. Here is Physicsnerd’s rebuttal to go along with mine:
Re: The Etp Model, Q & A Pt. 9
Postby Physicsnerd » Wed 01 Mar 2017, 23:33:28
shortonoil, “No. Entropy always increases in any system.”
dcoyne78, “If you had said “in any isolated system”, you would have been right.”
delta-S = delta-S(system) + delta-S(environment) >0 for natural processes. Nice nit-picking Dennis, but no.
delta-S is the change in entropy which is positive. You may have a negative “change in entropy” for the [delta-S(system)], which is not ‘negative entropy’. It is that portion of entropy that is negative.
dcoyne78, “Does this make it clear why, “No. Entropy always increases in any system.”, is incorrect?”
No, negative entropy is something you insist upon, somehow someway.
——–
So, Dennis, please stop further embarrassing yourself on this issue.
Meanwhile, I have a couple of questions for you. Judging by the timestamps on your thousands of comments and the shear number of graphs you churn out, you obviously put an incredible amount of time and effort into your online presence. Do you have another job or do you do this for a living? Who pays for this site?
Hi Cunning Linguist,
I specifically said the change in entropy is negative.
dS/dt=Q_dot/T+ sigma.
In the problem cited Q_dot is negative, T is absolute temperature and must be positive and sigma will be less than the absolute value of Q_dot over T in this case.
The change in entropy will be negative for the hot object. System plus surroundings (an isolated system) will have a positive change in entropy as the heat flow into the surroundings increases the entropy, irreversibilities (sigma) ensure that the isolated system will have a positive change in entropy.
See https://en.wikipedia.org/wiki/Entropy
and see section on Entropy of a System
As an example, for a glass of ice water in air at room temperature, the difference in temperature between a warm room (the surroundings) and cold glass of ice and water (the system and not part of the room), begins to be equalized as portions of the thermal energy from the warm surroundings spread to the cooler system of ice and water. Over time the temperature of the glass and its contents and the temperature of the room become equal. The entropy of the room has decreased as some of its energy has been dispersed to the ice and water. However, as calculated in the example, the entropy of the system of ice and water has increased more than the entropy of the surrounding room has decreased. In an isolated system such as the room and ice water taken together, the dispersal of energy from warmer to cooler always results in a net increase in entropy. Thus, when the “universe” of the room and ice water system has reached a temperature equilibrium, the entropy change from the initial state is at a maximum.
Entropy is always positive, but change in entropy for a system that is not isolated can be positive or negative.
When something warms up, the change in entropy is positive (cold glass of water),when it cools down (hot cup of tea) the reverse is true.
Note that you try hard to twist words,but that is all it is,
I have never said entropy is negative,only that the change in entropy of a system that is not isolated can be negative. For a system that is not isolated the change in entropy can be positive or negative.
Absolutely correct that for an isolated system the change in entropy will be positive.
Not all systems are isolated.
Dennis,
Your little physics experiment, which you are still incorrectly interpreting by the way, has nothing to do with the oil production process. All processes experience rising entropy. This is what the second law of thermodynamics is all about. Irreversibilities build in the oil production process (the oil production process is not reversible). This is rising (positive) entropy production. The Etp model correctly uses the Entropy Rate Balance Equation for Control Volumes to derive the rate of entropy increase in the oil production process. This tells us the total energy used to produce, refine, and distribute oil and it’s byproducts.
How do you answer this question: “Is the oil production process reversible?” If you say yes, you are obviously just wrong. If you correctly say no, then what the hell are you blathering about? Your experiment in no way relates to the oil production process. We are talking about an irreversible process and properly applying the second law using the Entropy Rate Balance Equation for Control Volumes to analyze it. What is wrong with that?
And you failed to answer my other questions:
Do you have another job or do you do this for a living?
Who pays for this site?
These are fair questions, Dennis. Please answer them. Thank you.
So by your lack of answer, I will assume that you concede that you are a shill and this site is paid for by big oil.
The site costs very little to run, it is paid for by me since May 2016 and before that by Ron Patterson.
I don’t imagine the oil industry pays for many peak oil blogs. 🙂
I have never claimed that there are no irreversibilities in the oil production process, however Hill reverses the sign of heat flow to make the irreversibilities have a positive sign, one cannot change the sign on the heat flow without changing the sign on both sides of the equation, a very basic algebraic mistake by Hill, either the man doesn’t know algebra or doesn’t understand thermodynamics.
SK has this right, Hill does not.
Do the math!
Hi SK,
Thank you for this great post. Sorry about the errors, the equations have been corrected.
I did take two semesters of classical thermodynamics as an undergrad ME major and one semester of Heat Transfer, I switched to physics in my third year and have a semester of statistical mechanics as well. No doubt you have forgotten more thermodynamics than most of us here have ever learned.
You bet that Hill analysis is off. They come up with a 50% loss of energy, well to gas tank and Hall calculated a 60% loss.
GF,
Any references to that work by Hall?
Here is one reference.
http://www.sciencedirect.com/science/article/pii/S0301421513003856
GF,
Where in that paper you linked to says 60% loss of energy from well to gas tank?
The paper has a table that lists EROI (oil and gas) for some areas which spans from 8 to 65.
Reread the paper. Even shown in a pictograph.
Oh, no. The work of Charles Hall is littered with mathematical errors which invalidate his conclusions. It’s just all wrong.
I wrote a blog post on this yesterday:
http://bountifulenergy.blogspot.com/2017/03/dr-charles-hall-is-still-totally-wrong.html
-Tom S
^ I realize it’s different, incidentally, just that it made me think about that and losses in general.
True, the petroleum industry does not use 57% of the petroleum. There are a lot of external inputs to the drilling field, the refinery, and distribution. Coal and natural gas provide much of those inputs as well as petroleum products. Total energy input is about 40 percent of the oil energy if one excludes the transportation system.
GF
Do you have references (like real world examples) to your claim?
Rune, we went through that a few months ago on this site. I provided examples and real world data with calculations then. Will see if I can find it again.
Caelan,
In recent days oil companies have reported their results and many comes out with an LOE of $7-$8/bo (and some less).
This by itself shows how far off the estimate of an EROEI of 1.75 is.
Understood, Rune, thanks.
With a thermal efficiency of about 20% (like for a good chain saw) one barrel of crude oil (6 GJ; that is 6 000 000 000 Joules) would provide about 335 kWh of useful work.
One man doing hard manual labor may produce about 70 W or 0.07 kWh in an hour.
So one barrel of crude oil has the potential to provide for about 4,800 hours of hard manual labor in the woods.
That barrel of crude oil costs now about $50/b could substitute for about 4,800 hours (more than 2 years) of manual labor. That amounts to about $0.0104/hr (yes a little more than a penny for an hour!)
OK, since crude oil is not used, but its derivative gasoline, which after refining and distribution would end up costing the user about $0.02 to substitute for one hour of hard manual labor.
How many are now prepared to outbid crude oil (rather its derivatives) with his/hers manual labor, that is undercut crude oil on price?
I don’t know Rune. We had to build a giant technological based industrial complex to even produce that chain saw and support the further use of that saw. Also with the faster production had to go faster and vastly more complex means of transport and processing as well as distribution. That is a lot of energy just to get more forests cut down more quickly. And of course to have a need for faster production we had to build a bigger market, meaning a vastly larger population with increasing demands.
One thing leads to another and the need for something like the powered chainsaw, automobile, etc. was determined by growth, which was fed by the power of fueled machines. Fossil fuels allowed a limited but uncontrolled feedback of growth in population, economics, industry and agriculture. The whole system is now slowing down, but is so interdependent that no piece can easily be removed or replaced without debilitating the system we depend upon for survival.
We have committed a paradox and are now a biological system caught in a technological diminishing loop. Or to put it more simply, we have boxed ourselves in and the box is growing smaller (while we grow bigger).
So when one adds up all the work and costs of making and running a techno/industrial civilization, is the chainsaw actually using less energy and costing us less? Maybe the cost is just distributed across all of society.
And your point is?
That chainsaws are dependent upon a large technical industrial complex society, while manual labor is not. That means that direct energy comparison is not valid.
Also the side effects of fossil fuel energy coupled to an industrial technical society are to force a probable dead end.
Hi Gonefishing,
One interpretation of Rune’s comment on oil and chain saws is that oil is a very useful product which can save a lot of human labor. All of the infrastructure that makes the us of oil and gas has already been installed and the only extra input is ongoing maintenance, all included in the cost of a gallon of gas along with extraction, transportation (pipeline or rail), refining, and distribution. The cost of an hour of human labor is fairly high (especially in the OECD) so people are willing to pay quite a bit to save on labor costs.
In short, it is unlikely that we would see the demand for oil decrease to the point that oil prices will be low if the market is not over supplied. Eventually prices will need to reach a level where the cost to produce the most expensive barrels of oil required to meet demand is equal to the price of a barrel of oil. If not, a rational business will not produce that oil in the long run because their profits would be negative and eventually they would become bankrupt.
This is just one of many reasons why the ETP Model is incorrect, along with the reasons already given by SK, Rune, and you, among others.
How many are now prepared to outbid crude oil (rather its derivatives) with his/hers manual labor, that is undercut crude oil on price?
Why would you want to? Gasoline powered chain saws are finicky, smelly, dirty and noisy. I myself have been using Lithium-ion battery powered tools including saws for a few years now. I built my first solar charger for my power tools about five years ago. and technology has moved forward since then. I don’t own a chain saw but if I had a need for one I would get something like this…
http://sustainableus.org/2016/05/27/a-real-life-solar-powered-chain-saw/
A Real-life, Solar-Powered Chain Saw
I took a risk, and I think it paid off. I have an electric car, and a cordless electric lawnmower, and fully-functional battery-powered construction tools. But a chain saw? I was pretty skeptical, but I was also intrigued by the potential advantages—push-button starting, light weight, not having to mess with gasoline mixes, no finicky carburetors to keep adjusted. So I spent some time watching YouTube videos of electric saws, and decided that one of the larger ones might indeed work as well as my Jonsered gas saws.
So I took bit of a gamble, and ordered one from Amazon. It’s an 80-volt, 18-inch Greenworks saw, and comes with a 2-amp-hour lithium-ion battery and a 30-minute quick charger. I also ordered a second battery. Three days ago the package showed up on the porch, and I have to say, I’m really impressed with it so far, so much so that I’ve already made arrangements to sell the gas saws.
Ok, before I go on, it’s obviously only indirectly “solar powered”, because I charge the batteries at home from my net-zero solar set-up. But that was one of my goals– to further reduce my fossil-fuel use. When I charge them at home, they are indeed solar powered.
Disclaimer: I do have an agenda and it is to promote alternative energy, especially solar and batteries, because fossil fuels ain’t all they’re cracked up to be! (pun intended) 😉
Fred, thanks!
Some day in the future we will have to do with much less fossil fuels.
And by then you are prepared to outbid the competition. 😉
”And by then you are prepared to outbid the competition. ?
This is why the leaders of countries such as Germany and China are so dead serious about force feeding their domestic renewable energy industries.
When fossil fuel prices start going up, up , up, and they must, eventually, due to depletion alone, ……….. Germany and China will have a HUGE competitive advantage.
Hi Fred,
As you well know, I’m as big a fan of renewable energy as anybody, but excepting light duty use by a home owner, battery powered chainsaws just aren’t there yet.
If you buy a GOOD i c e chainsaw, you can reasonably expect it to run for at least a thousand hours before you have to do more than change the spark plug plus routine maintenance and you can expect it to start first pull just about EVERY time.
Ninety nine percent of the saws sold are homeowner junk. All the cheaper models sold by the major brand names are home owner junk. After spending a number of hours getting up to speed on current makes and models by talking to people who actually use them on a daily basis, I bought a new brand X model for five hundred bucks recently, which particular model is considered by local woodsmen to be the cheapest saw they will even THINK about buying.
Incidentally I own the following battery powered tools, and LOVE them, and wouldn’t even THINK about being without them. Three drills, various capacities including hammer function, screwdriver, drywall screw gun, reciprocating saw, rotary saw, right angle grinder, rotary cut off tool for thin metal, flood light, flash light, half inch square drive impact wrench, fuel transfer pump which could also be used to pump potable water if thoroughly cleaned. I also have a trailer mounted winch that originally came with a hand crank which I modified to so I can operate it with my biggest battery powered drill. One hot battery will pull a full sized pickup up onto the trailer no problem at all with some charge to spare.
The biggest problem with them is that the manufacturers need to be locked up in a pit someplace, without any sanitary facilities, until they agree to standardize the battery interface. I can get along fine all day with two to four fully charged batteries using any of these tools.
Out in the woods , I would need at least six or eight batteries just for ONE tool, and my productivity would be miserable.
OFM, “Out in the woods , I would need at least six or eight batteries just for ONE tool, and my productivity would be miserable”…
Just curious, are NiCads, Li-ion or something else?
The batteries that come with electric chainsaws are lithium ion at least among the ones I have checked out. And none of them are powerful enough to run the saw more than about an hour, at the most, unless doing only extremely light work such as sawing up small limbs to stove length, to the best of my knowledge.
But finding an accurate and objective performance test run by somebody out to discover the truth, rather than to either promote or badmouth battery powered chainsaws is not easy.
“And none of them are powerful enough to run the saw more than about an hour, at the most, unless doing only extremely light work such as sawing up small limbs to stove length, to the best of my knowledge. ”
That is correct. I have such a saw. For work in garden ok, for real work in the woods not so useful.
Out in the woods , I would need at least six or eight batteries just for ONE tool, and my productivity would be miserable.
Now this is exactly where solar can really shine! 😉
As I mentioned up thread I built my own portable PV solar charger for my power tools years ago and could build a much more powerful one for a lot less today.
Today I would probably use lightweight flexible panels.
BTW, for JN2, I have switched almost all my battery packs to Lithium-ion, they even fit my old tools that used NiCads.
Back atcha Fred,
I am willing to believe that within five to ten years, I might be able to buy a nice portable solar array that folds up and is easily transported that would be adequate to charge chainsaw batteries fast enough that I could work with only two or three batteries.
But it won’t be cheap, compared to a gallon of gasoline.
And of course in case of real need, I could just saw for an hour or so on lots of different days, and get in enough wood that way. But my woodlot is a half a mile from the house…….. and to reduce the use of gasoline or diesel fuel to the bare minimum, I would have to walk there and back, which means more time per cord.
Anybody thinking about homesteading will do well to buy their property in one tract if they can do so without paying a big premium. But our house is built on only a couple of acres given to Daddy as a gift from his Dad. All our other land is at least a quarter mile away, and was bought piecemeal as it became available.
The separated tracts are definitely an issue, but once you get an electric truck…
I am guessing they’ll become widely available circa 2022.
But it won’t be cheap, compared to a gallon of gasoline.
I dunno. I you’re really working 8 hours a day, and using it for full time work for the majority of the year, I suspect that it would pay for itself pretty quick.
Have you looked at leaf blowers? I’m curious – I’m an urbanite, and gas leaf blowers are a real blight!
I have outlets strategically located outside, and thus use a 120 volt ac leaf blower, which is still pretty loud. For another fifty bucks or so, a gasoline powered model could be made to run about as quietly as an electric model.
The solution for NOW would appear to be stricter local noise ordinances, lol.
I’m hoping batteries continue to get better and cheaper as fast as predicted by optimists.
But I don’t yet see any real signs that they will get much cheaper SOON, at retail, for homeowners, because the manufacturers make them so they are deliberately not interchangeable with generics.
Once such ordinances are enacted in a few large cities, you can bet that super quiet ice models will be in the stores within a year. ICE engineers know all about how to make really good mufflers, lol.
For another fifty bucks or so, a gasoline powered model could be made to run about as quietly as an electric model.
Too bad they’re not available now.
Well…any recommendations for effective electric models?
Heh. Lack of standardized batteries is irritating, isn’t it? It’ll solve itself after a while.
Productivity with swappable batteries is plenty high enough. Though carrying a pile of batteries may seem annoying, it’s better than carrying gas cans and oil cans.
Productivity out in the field working with today’s batteries SUCKS big time.
That’s why hardly anybody at all uses battery powered tools for production work except on job sites where there is ac power for charging, and where the tool is used intermittently for the most part, if it consumes a lot of power. I can and do use my battery powered circular saw aka skilsaw when doing repair work, or a little work out in the field.
I have never yet met a woodsman who owns a battery powered chainsaw, a farmer who owns a battery powered tractor, or a lawn and garden guy who uses a battery powered lawn mower, but there are some used commercially. They use them in quiet zones, such as hospital grounds, and get paid extra for doing so.
A pile of batteries is a pain in the ass compared to a gas can, which can stand a lot of abuse. All the care mine ever gets is to strap it down in the truck. It’s often left sitting out in the woods, or on the back of the truck right thru a week of rain or snow.
I’m willing to believe batteries will eventually be good enough and cheap enough to displace small ice engines in trades work but my bet is that such batteries are at least five to ten years away.
Question for any body that knows:
Is there any real NEED to EVER decommission a wind farm?
I can’t see it myself. Now a few older wind farms might need a complete overhaul, with new towers and turbines, but unless the land becomes so valuable the wind farm needs to go for that reason, WHY should any wind farm ever be shut down permanently?
I’m thinking the final line in this link is bullshit.
http://www.caller.com/story/opinion/readers/2017/02/27/city-learning-mitigate-negatives-wind-energy/98489488/
There might not be any need for a new coal or gas fired power plant in the same or a nearby spot when an old one is finally worn out and scrapped , but with the roads and transmission lines, etc, in place, the permits all grandfathered, etc, WHY would a wind farm ever NEED to be permanently closed ??
Hi all,
The post is also up at peakoil.com.
http://peakoil.com/consumption/on-the-thermodynamic-model-of-oil-extraction-by-the-hills-group
Re: On The Thermodynamic Model of Oil Extraction by Hill’s G
“Is that you Loren Soman?” ~ Rune Likvern
Where is Hill in this dialogue? I await their response.
Hill’s responses, along with others’ like Ghung’s, are on the mirrored-article site, Peak Oil News.
Sample:
Hi Caelan,
In the Hill’s group report on page 8 near the top of the page Hill states that because the heat is flowing out of the system he reverses the sign on sigma. This is the reverse of the usual engineering convention where heat flows into a system (heat gains) are positive and heat flows out of the system (heat losses) are negative.
We could just as well claim that dS/dt always decreases and change the sign on the second law of thermodynamics!
Then the folks at peak oil dot com including shortonoil (Hill) claim that the correct equation by SK is wrong because he has used the standard definition of dQ/dt. Note that Clausius would have defined entropy with the reverse sign if this convention had been used and the Second Law of thermodynamics would state that for an isolated system entropy always decreases.
I would never say that but that is what Hill’s equation near the top of page 8 of his report implies by reversing the convention for heat flow in thermodynamics.
Its like up is down in that Wonderland. 🙂
They have posted here before. I suppose they know they have little to do with the well informed oil experts that contribute to this blog comments, so they concentrate their artillery on the Peak Oil News mirror.
Looks like the US was a NET Nat Gas importer for 2016.
2017 Who knows? How much will price rise with increased exports?
http://peakoil.com/consumption/the-us-is-transitioning-into-a-natural-gas-exporter
Longtimer
The just-released data point from Cabot regarding EURs from their most recent (21) wells in Susquehanna county is one of the most significant pieces of info that I’ve encountered in 10 years of following this so called Shale Revolution.
Cabot claims future production of 4.4 Bcf per 1,000 of lateral.
As they now project 8,000′ laterals the norm in the future – present cost $8 million per – Cabot expects to ultimately get 32 Bcf +/- for each well.
That is so astonishing as to strain credulity but for the short production history of these wells where 1 Bcf per month has already happened.
Described another way, in terms of energy produced comparable to barrels of oil (perhaps relevant to this thread’s topic), Cabot can, right NOW, spend 8 million bucks and produce the energy equivalent of over 6 million barrels of oil.
The ramifications of this are very big.
Yes it would imply, if true an oversupply of natural gas to the point that natural gas prices will be very low, perhaps to the point that producers will not be able to make any money. Perhaps they can export the natural gas, but this sounds like hype. The wells may be very good, but there may be very few of these.
in wells I have a Working interest in, my first 10,000′ lateral took 103 days, the last two less than 35 days. Went from less then 1000 lbs per 1000 feet of lateral to 2000-3000lbs of sand per 1000 feet of lateral. Went from $16,000,000 per well down to less than $10,000,000 per well, EUR have gone up from 2000MBOE to 3000MBOE in less than 2 years. I have no idea if we will produce 3,000MBOE but I have wells, that at current prices, that i get my money back IN FULL in less that 6 months with combined WI and RI. I hope Dennis’s predictions down thread of $100-$125 in 2020 are correct.
Could not tell if his prediction was a depression due to higher energy prices or some other cause.
My oldest scoop well, now has produced 6BCF+ 277,000BNGL + 49,691 BO from a 4500′ lateral with a 3,200,000 lbs frac. Continues to produce just under 2000MCFD & 85BNGLD & 8BOPD. This section will have at least 9 other wells that will have laterals at least a mile long with twice the frac size. I can see how future EUR could double the expected EUR from this well.
I can now say my oldest Woodford OIL well in scoop continues to produce at a rate almost 50% higher than the average Eagleford well, per Dennis’s EUR published on this site, at month 21. (150BOPD & 1900MCFD) operator does not break out NGL on this well. Been very steady with no oil production decline now for 6 months. Jury still out on EUR, but i ain’t complaining.
When subsidies are removed from wind and solar, just a matter of time, I expect to see higher prices. The oil and gas naysayers will be proven largely wrong within any time frame as measured by my expected time on this earth.?
Dennis/TT
The 2/26/2017 post over at RBN has what may be the most concise description of what is currently driving unconventional production today.
A ‘must’ 5 minute read if one wants to understand how operators can continue to grow production, add rigs, and analyze future ramifications.
Drilling one mile laterals in a day is not the norm, but it is happening more regularly.
The diversion technology has advanced so swiftly that refracs, as well as new completions, will turn earlier projections aside.
A small outfit, Travis Peak, is set to drill Pennsylvania’s first 15,000′ lateral (The Purple Hayes from Eclipse, 18,500′ lateral, is in Ohio).
At a depth of 10,000 feet, this effort – if successful – will dramatically lower production costs for future wells with this configuration.
I realize that this is a peak oil blog, but you folks may be shocked at the accessible hydrocarbons looming in the future.
The Mancos shale, recently assessed at 66 Tcf by USGS, covers an area the size of Dunn county, ND.
Its 4,000 foot thick pay zone has high clay content, but the fuzzy heads are working on that with the trend towards cryogenic (gas) as a carrying/frac’ing medium.
You guys ain’t seen nothing yet.
Still waiting to see them turn a consistent profit.
And this is just in the Marcellus/Utica, correct? In a stranded basin?. Every other shale gas play is sucking wind. Barnett, Fayetville and Haynesville. I really would like to hear one of these companies state when their wells actually pay out, and now that we are 8+ years into this, we can quit talking about EUR and start talking about true reserves (what they pull out of the ground)
Four years ago they were hedging into a $4+ gas market and banks were happy to loan them money. Not anymore.
These companies financial expertise better catch up with their technical expertise
RT
A lot of people are skeptical about the financial aspects, to put it mildly. As is my inclination, I focus on operations, not the business side of things.
FWIW, every 1 Bcf at 3 bucks/mmbtu is $3 million gross revenue. Enno’s site, under cum production, gives a rough view of money coming in.
Obviously no wheres near net, but significant numbers just the same.
As far as Cabot’s EURs, Susquehanna county may hold the most productive resource potential for gas in the USA.
There are other area with great rock, but 4.4 Bcf/1,000′ is remarkable.
Coffee
I know you look strictly at production and operations and I know how to calculate the value of 1 BCF of gas. I will just say, the world is full of insolvent companies with significant revenues.
You mention enno’s site. The best for evaluating the ACTUAL production. AS opposed to the ESTIMATED production they like to tout on their P4 reserves. Looking at that site, and comparing to all of the quarterly reports and power point presentations, why would you ever believe what these companies are selling? Shouldn’t you wait to see the actual production? My guess is the numbers from Enno’s site don’t match the numbers these companies touted in the last 5 years.
I will just add, at some point, if you have multiple rigs running and you are making wells for every well they drill, shouldn’t you be able to finance your capex through cash flow as opposed to debt?
Production volumes reported in quarterly reports are correct and do not contradict Enno’s data.
Projections and estimates in presentations are a different thing
I understand that. My complaint is with the EUR numbers that are thrown around in their investor presentations. My point was to compare those numbers to the actual numbers produced.
Yes, EURs in presentations are generally well above actual average numbers
RT
Pulling data from Enno’s site (amazing source for info), looks like a Narden well from Warren ranks #933 out of ’bout 7,000 PA producers.
This well produced 5 Bcf, grossing $15 million @$3/mmbtu.
So, way over 900 wells in Pennsylvania alone have produced above that amount.
(Ohio’s Utica had nearly 60 wells produce over 1 Bcf in 3rd Qtr., 2016).
More pertinently, perhaps, is the most recent wells from Cabot with their preposterously high EURs …
The pads King, Ferguson, Housenick, Halliday and Bolcato are the sites for these newest wells.
Flow rate is running from 18 to 28 MMcfd for many with cums at 1.5 to 3.7 Bcf with the ‘oldest’ online 138 days.
The fact that Cabot plans on implementing these latest completion techniques (as, indeed, are operators everywhere) should give indication of where future output is headed.
Technical wonders aside, or how much money you might be personally making from royalty interest (free and clear of all costs), focusing on a few good wells out of tens of thousands of lousy wells is nothing more than self-entertainment. In the mean time, Rome is burning.
Four out of the five or six top shale oil producers in the Bakken (Hess, Continental, EOG, Whitting) lost $9B in 2016, the two biggest producers in SCOOP/STACK (Continental and Marathon) lost $2.5B and Cabot, in the Ap Basin shale gas play, lost a paltry 417 million, 293 million in 4Q16 alone, this in spite of much higher gas prices and mucho grande IP90/EUR’s.
Giddy up!
Coffee
What about the rest of the story?
All the other wells? The costs? and ultimately, the profitability of the companies?
Every producer who has been in this business for longer than a couple years can point to fields, plays, prospects, etc that were sold on completing every well you drilled. They all produced oil or gas and they all generated cash flow. Heaven help you if you ever purchased an interest in one of those deals.
The shale plays are a bigger, more sophisticated version of those promoters from years past. Pretty maps, EUR and cash flow analysis for those investors presentation.
You can not discuss production and reserves without discussing costs and profitability.
RT
I agree wholeheartedly that economic viability is crucial lest this whole Shale stuff turn out to be a horrifically destructive Ponzi scheme, as many seem to claim.
Lottsa factors involved, most of which we outsiders barely even know.
But, RT and Mike, I’ll post in the coming days some observations on Enno’s site that may offer some context on the numbers his data projects.
Specifically, when one includes ALL the wells, as Enno does, the multitude of dogs gets weighed somewhat similarly to the superstar wells.
With the vast expanse of the Bakken (3 to 6 thousand sq. miles) along with the 70,000 + square miles of Marcellus, a great many wells were drilled in rapid fire fashion to simply protect leasing.
There is a ‘lot’ more to the story, but – going forward – these unconventional operations will continue to improve both financially and operationally.
The biggest losers by FAR are the Saudis, and they sure as shit know it.
Mr. Coffeeguzz, I am disheartened once again how easy it is for you to completely ignore the failed economics of unconventional shale extraction and instead to focus only on what interests you and what strikes your personal fancy. I appreciate your passion for gadgetry and technology in the oil and gas business; I work with it every day and it is indeed very cool. I fail to see, however, what that has to do with peak oil concerns, on a peak oil blog, other than you wish to prove to everyone you have special insight into our future, and we need not worry.
I gave you a snapshot example this morning of a half dozen shale oil and shale gas companies that lost over 10 billion dollars in 2016, those horrific losses in spite of higher product prices, greater so-called “efficiencies,” lower finding and production costs and new and improved “technology.” It went plum over your head. Mr. Hightower is absolutely correct; not you nor anyone else can constructively discuss production and reserves without also addressing costs, lack of profitability and the dire current financial condition of the industry you are so enamored with.
You can, I suppose, but it is a deep form of denial and I wish better for you.
Private enterprise in America is failing at extracting high cost, high decline shale resources. Costs were now headed back up, product prices back down, and the big ugly beast in the room, debt, never goes away. The interest meter never stops whirling.
What matters about our energy future is not barrels and mcf’s. Its dollars and cents.
Mr. Roughneck
If a baker sells his loaf of bread for 2 bucks per instead of 6, if a builder is getting 100k for a new build versus 300k, the negative consequences would be immediate and catastrophic.
This, in essence, is what has happened to all in the hydrocarbon production industry.
While the bakery model is somewhat inadequate to equate with the technical innovation in E&P, house building via 3D printing may offer a rough analogy.
If, as is currently possible on a prototype basis, 2 story, custom designed houses can be built in a few days with minimal labor, the existing home building industry would be turned on its head.
If, hypothetically speaking, there was a powerful government entity that felt threatened by this new technology, that absolutely needed the revenue provided by its own house building activities, it might very well try to destroy the fledgling upstarts.
OPEC NEEDS the sustainable revenues from their oil and gas.
The fact that two years of low pricing has financially hurt the shale guys should be no surprise as that was the goal all along.
Folks such as yourself, RT, Shallow and many, many others have been hurt in the process.
Hasn’t worked.
These guys have already started to recover and the enabling factors included all this described innovation coupled with a ferocity to get ‘er done that you yourself have witnessed for a lifetime.
This method – producing hydrocarbons via one mile, four mile long laterals, fractured with unimaginably large amounts of proppant – is the main mode of O&G production in the US for the foreseeable future.
Thanks for the response but I have no freakin’ idea what you just said. If folks here on POB like your chili, I can’t help anymore.
Except to say this; it has been a 30 month ploy of the US shale oil industry to blame OPEC for its woes. The facts are that the shale oil industry borrowed upwards of half a trillion dollars of upstream and midstream capital to put 4.8M BOPD on an otherwise balanced world oil market. The price collapsed, accordingly, by 65%. In the face of this overleveraged oversupply, a whacked out federal monetary system that funded it, and the lifting of a 45 year old export ban, OPEC then, and only then, began increasing its production, in the name of market share. I would have done the same thing. The shale oil industry shot itself in the foot.
http://peakoilbarrel.com/opec-crude-oil-production-charts/
Five months ago OPEC cut its production. In spite of this leg up by OPEC just 5 or 6 shale oil companies I picked this morning lost $10B in 2016. There are a bunch more that lost just as much.
Now, the shale oil industry is whining about cutting regulations; that’s the fix de jour. Regulatory compliance costs in the oil and gas industry are negligible, two bits per incremental barrel, at most. When that doesn’t help it, what’s next?
I know, import tariffs. That should do it. But maybe not; there is still over 250 billion dollars of unpaid debt, this in spite of the shale oil industry having crapped out already on about a $100B (Haynes and Boone).
Personally, I think that OPEC is not stupid. Its now done its homework on these ridiculous EIA reserve estimates, and debt, and no longer considers the US shale oil industry a threat. So, it has given it yet another long rope to hang itself. It knows the Bakken and the Eagle Ford have peaked, the shale oil industry cannot manage its massive debt much longer, interest rates have to go up and as soon as the Permian ends up like other shale oil plays in America, there is nothing left; the last of America’s remaining resources are gone. Then OPEC has us right where it wants us, back on the teet, big time, for the last time.
That’s how greedy and short sighted the US shale oil industry is. It sold us all down the river.
“The biggest losers by FAR are the Saudis, and they sure as shit know it.”
Coffee,
There is no beef in this narrative. Due to agreement in petro dollar recycling from the Nixon era, House of saud and Wall str. are connected at the hip. One goes down, the other goes down too. It is mutually distractive agreement. That is reality. There were other policies in previous administration where they tried to play Saudis against Iran with that Iran nuclear deal. But Iranians are not biting.
Bottom line is very simple too much debt and not much oil to keep economic system lubed.
Hi Coffeguyz,
The important question is the cost to drill the well rather than how much time it takes. If they can complete the well in 5 minutes, but it costs a billion dollars to do it, the bottom line is not helped, it gets worse. Well cost is the key, I don’t care how long it takes.
Come on Dennis you are smarter than that. TIME = MONEY in the oil field.
Hi Texas Tea,
Yes time=money. My point is that it is the money that matters. If the well costs twice as much, to complete in half the time, will the company come out ahead?
As I said whether it is 1 day or thirty days does not matter, only total cost.
Maybe Mike or Shallow Sand can correct me, I am not an oil man.
my biggest complaint is those on this venue who constantly preach how “renewables” are going to get cheaper and more efficient but at the same can’t wrap there heads around the same thing happens in the oil patch. The only difference is my business is not subsidized and the market place has made its decision. With out government coercion renewables will drop like a rock in terms of addition to our energy mix. When they are ready for prime time it wont take government involvement to roll them out. IN the mean time I got check to cash?
>> renewables will drop like a rock <<
Nope, wind and solar are already cheaper (unsubsidized!) than gas, coal and nuclear new builds. The PTC (production tax credit) for wind is being phased out to zero by 2020, by which time wind will be even cheaper and more competitive.
PS Don't you live in Texas? Haven't you seen the wind numbers? And solar insolation averages 6,000 barrels per square mile per day…
I live in the real world, care to join me?
http://blogs.platts.com/2015/07/16/burn-baby-burn-north-american-natural-gas-generation/
For reasons unknown it takes me 36 or so hours to see a new post. Y’all have gone crazy over this one.
Suggestion to author guy seeking anonymity. Commit yourself to the idea of “I am nobody and I’m gonna stay that way.” It is all much easier then.
EROEI stuff isn’t going to tell us anything because the measurements are all undefined. Always seemed annoying when doods tried to quantify energy release from breaking a H C bond and then neglected to address the energy consumed breaking the next bond.
Overall big picture stuff about thermodynamic modeling: All crap. Safely ignored.
For reasons unknown it takes me 36 or so hours to see a new post.
It is due to a WordPress update issue and it affects everyone. Try refreshing your browser when you access this site and then refresh it again when you look at the comments. It’s a bit annoying but takes less than 36 hours…
thanx
You might want to clear your browser’s cookies/history as well.
POB has been acting funny lately.
Hi Fred,
So If I update Word Press will this problem be better or worse?
I am a little skeptical that I can successfully undo any updates.
For reasons unknown it takes me 36 or so hours to see a new post.
Watcher, you have to refresh to see any new posts. They will never appear unless you refreshes your screen. But when you do refresh you will see all new posts immediately. You will not have to wait 36 hours, you will not have to wait one minute, they will appear immediately. Damn, this is not rocket science.
This is not like email, where new posts appear without any refresh whatsoever. On PeakOilBarrel, the screen will remain completely static unless you refresh. It has always been like that. This is not anything new.
Dennis, you must update. The updates will keep coming and coming. They do not cause problems, they fix problems. Don’t let Watcher’s inability to refresh his screen become your problem.
Hi Dennis, I have a Word Press website of my own i only update it when WordPress sends out an update. I’m a strong believer of the “If it ain’t broke don’t fix it” mode of doing things. Like I said to Watcher while it may be an minor annoyance, all you have to do is refresh your browser to see new post.
It has always been like that. This is not anything new.
Actually this is something new because I have been coming here for a long time and all I had to do was access the POB url and I would see all new posts, it has only been in the last month that I have had to refresh my browser to do so. Others have also noticed and commented on this.
In any case you are right, refreshing a page isn’t rocket science and it certainly shouldn’t take 36 hours to see new posts!
Cheers!
Yes, these days, I have to hit refresh each time I come here. And I have to refresh each post to see the latest comments there. It isn’t sufficient just to refresh the first page I land on at POB. To see the latest comments for any of the posts, I have to refresh that page. So nothing automatically refreshes anymore. Each page requires a refresh.
Hi Boomer II,
Ever since the site went down about a month ago, there has been this problem. I may pay a little more next year for better service. Ron was less cheap than me about paying the hosting service, I tried to cut costs a bit.
Maybe I will see if people would make donations, I have wanted to avoid this, but my funds are not unlimited.
It’s not a huge hassle for me. I’m just letting people know that this seems to be the drill for everyone now.
Been acting a little squirrely for me also these past couple weeks.
Dennis, a guy who blogs on Marcellus gas asks for 12 bucks a year donation and it seems to work for everyone.
I’d be happy to kick in for all the effort you all put in.
Thanks Coffeguyz,
If I do it I would try to make it voluntary so we can test out the free rider hypothesis 🙂
36 hours? They don’t call him watcher for nothing!
“Overall big picture stuff about thermodynamic modeling: All crap. Safely ignored.”
Yeah, that was my conclusion a long time ago. Still true.
The Etp report says that
Report# HC3-433
Version 2, March 1 2015
“Depletion: A determination for the world’s petroleum reserve”
From The Hills Group
”2012 is the point where the petroleum production system (PPS) becomes a larger user of the energy that comes from crude oil than is the non-energy goods producing sector (NEGs) of the economy.”
Note this is a result from the report, not a claim or aworking theory put to test.
Page 36
” The extraction, processing and distribution energy costs for 2012, when summed, equal 70,000 BTU/gal; which is what the ETP model predicts.”
Why does this not show up in the financial statements from the oil companies?
” It therefore follows that average extraction costs are close to sale price.”
Page 49
The Etp report has no links to other energy sources like natural gas, coal, nuclear, hydro and other renewables.
The give away is that it in 2012 took one barrel of oil (or its equivalent in energy) to deliver one barrel of oil to society. Now society may even receive less than 1 barrel.
Since the Etp model only looks at petroleum and the oil companies primarily use diesel this means that the oil companies sells 2 barrels of crude oil and consumes one barrel of diesel in the process.
2 barrels of crude oil sells now for about $50/b or $100 for 2.
1 barrel of diesel retails now for about $100/b.
In other words the Etp model describes a system whereby there is no financial surplus after energy consumed in the process is paid for.
The consequences of the Etp model is that the financial costs of energy alone is about $50/b.
I have looked through several financial statements for Q4-16 and total LOE (Lease Operating Expense) which includes costs for energy ranges from $4 – $10/b.
This also suggests, like other peer reviewed studies (which I linked to another place in this post, official publications (like the Sankey for UK I linked to earlier), estimates from Norway (which has good data which I presented in a post here on POB back in October 16) that the Etp model now is if off by a factor of at least 10 in its estimates for EROEI.
That also means that all the other predictions are heavily influenced from this, and that the Etp model predictive capacity by design is poor, but could on rare occasions get the oil price right right and then by accident rather than by design.
I got a link.
https://energy.gov/sites/prod/files/2013/11/f4/bandwidth.pdf
Page 19, scroll.
Refinery consumes 2.1 Trillion BTUs/Year. (2.1 X 10 ^ 12) Not at all clear how many barrels are processed on that page. Probably somewhere in link but lazy.
1 barrel of oil is 5.6 X 10^6 BTUs energy content. So 375,000 barrels worth of oil energy to run a refinery for a year.
Typical refinery flowthrough — Don’t seem to be a typical. 200K bpd looks easy and several.
So that 375K barrels for yearly refining energy reqd is gonna be maybe 2/365 of the energy in the oil itself, thereabouts.
“That also means that all the other predictions are heavily influenced from this, and that the Etp model predictive capacity by design is poor, but could on rare occasions get the oil price right right and then by accident rather than by design.”
That’s exactly what I think. Right now the ETP price for 2017 is close to correct, but that may have nothing to do with anything except random chance. I figure by some not too far future date it will be obvious the model is completely wrong.
Below follows data on Volve. Volve is/was an independent, small production installation which recovered about 64 Mb of oil which was its primary production.
Data on Volve from NPD and NEA,
Production per calendar day 2016 (Shut down Sep-16)
Oil: 9,249 b/d
Natural gas: 0.73 Mcm/d
NGL: 68 B/d
Energy consumables;
Diesel: 13.46 ton/d
Natural gas: 0.07 Mcm/d
Helicopters, supply and standby vessels; 5 ton/d.
(The Black columns at the bottom in the chart)
Energy content of consumable and products;
Diesel 43 GJ/ton
Energy content crude oil and condensate 5,73 GJ/b (as proposed by BP)
Energy content natural gas 39 MJ/Sm3 (sales gas)
Energy content NGL (LPG) 4,13 GJ/b (Based on LPG, 26 MJ/liter)
Energy consumption downstream Volve and until the products reach the consumer, 6% of what leaves Volve.
2016 estimated (flowing) EROEI ex platform: 15.3
2016 estimated (flowing) EROEI end consumer: 7.6
Note the facilities were shut down in 2016 while flowing EROEI was high (and certainly not 2.0).
Below a figure showing full life cycle gross energy extraction/consumption for Volve.
Full life cycle EROEI for society: 8.6 (Volve was a small (marginal) discovery.
ain’t much
As is always true of EREOI stuff, might want to call asphalt waste, since its energy is never extracted.
True, and also goes for other non-energy of oil and natural gas.
Counting gross Joules from source to end user at least gives a chance to compare apples to apples.
I had a closer look at the reservoir and production data for Volve for 2016.
The reservoir lies at a depth of 2750-3120 metres.
Volve was produced with water injection for pressure support.
Water oil ratio 2016: 4.8 (4.8 barrels with water per barrel of oil)
Approx reservoir temperature: 80 deg C
The energy following one barrel of oil from reservoir to wellhead:
Heat: 220 MJ/bo (provided free and of little/no use and requires energy to lower to 20 deg C)
Potential (lifting): 30 MJ/bo (inclusive flow losses and not adjusted for water injection)
Chemical (1 barrel of oil): 5,730 MJ/bo
Process/support systems, heli shuttling, supply and standby vessel: 410 MJ/bo pumped to storage.
In 2016 there was in addition about 13 Standard cubic meters of natural gas with each barrel of oil. 1 Scm is about 40 MJ.
Anyone willing to take a shot at an estimate for EROEI (ex platform and for oil)?
Hi Rune,
I get 13:1 if I ignore the Natural gas produced and 14.2:1 if both the energy from the oil and natural gas are produced.
Do you have a rough estimate of energy used in the transport, refining, and distribution of the petroleum, so that we could estimate the reservoir to fuel tank EROEI? I think in the past we found this is pretty low, maybe dropping EROEI to 10:1 or so?
Actually in the paper linked below
http://www.sciencedirect.com/science/article/pii/S0301421513003856
In figure 1 on page 142,they suggest for an EROEI of 10:1 at the well head, that EROEI drops to 2.67:1. If the investment for transportation infrastructure is included, the 10:1 wellhead EROEI drops to 1.26:1.
I found about 6% of the net ex the installation is consumed for transport from the installation (oil only as nat gas is pressured up before leaving the installation), refining and distribution. About 350 MJ/bo.
Hi Rune,
Wow, that is lower than I expected. Only 8:1 from reservoir to consumer and maybe 2:1 when transportation infrastructure is included (though I believe this last step is an overestimate).
How do you arrive to 2:1?
After the consumers receives the products it becomes hard to follow them in the value chain and see where they end up.
Hi Rune,
I linked a paper by Hall et al.
http://www.sciencedirect.com/science/article/pii/S0301421513003856
On Fig 1 they have 37.5% of energy going to transportation infrastructure so for reservoir to gas tank about 12.5% of energy in the extracted barrel is needed (using your data), assuming the 37.5% estimate is correct (I think this is too high) we have 100/50=2:1.
Sometimes people take the next step and consider the thermal losses in the IC engine an find an exergy return on energy invested, that is one way to drop the number from 8:1 to 2:1, but I don’t think that is a fair comparison (I have assumed 25% efficiency from gas tank to wheels).
Energy for transportation infrastructure is IMO part of society.
Figure 1 (a Sankey chart) could have been accompanied with more explanations.
It shows EROEI at wellhead of 10.
The refining step also includes byproducts which I assume also is non energy use (may explain why it is so high).
The transportation step likely includes all transportation from source to consumer.
The branch with losses (37.5 MJ) makes it confusing and this could be Carnot losses.
If so, this makes the chart a hybrid that looks at gross delivered along part of the value chain and splits it up into useful work and losses at the consumer step.
Hi Rune,
I agree. If the thermal losses in the IC engine are included it would make sense.
As society gradually replaces fossil fuel powered transportation with electrically driven transport these thermal losses will be reduced and as wind and solar replace natural gas and coal there will also be fewer thermal losses in the electric power sector. The ratio of primary energy consumed to exergy produced will decrease over time, maybe improving from 3.5:1 to 2:1 over the next 30 years.
One must include the refinery steps and inputs.
Press the links under the figure for more details.
The figure refers to minimum EROI to operate a society.
Few months ago I looked up some stuff regarding oil consumed for food transport. The findings were that for the US about 500K bpd carried food to grocery store shelves.
Quite a bit more was required for all aspects of agriculture to grow what was carried to the shelves.
I’d suspect same thing for EREOI stuff. A lot more energy reqd to extract from the ground and refine than transport it to the local gasoline tank.
Watcher
For the Volve case I looked at 400 people in total involved/employed with 400 cars were involved, 3 shifts a 50 persons and office support and supplier functions and that each car used 1,200 liter gasoline per year.
This showed a gross energy use of about 50 GJ/cd, which is about 1.5% of the use for operating the installation.
If this was increased by a factor of 10 (which should be very generous) the EROEI for society drops from 10.7 to 10.2 for 2015.
Yes, it is about boundaries. I have stuck to the boundaries that is most commonly used.
My post here was about energy balances ex the installation (or ex wellhead as some refers to it).
What prevents you from digging out the data yourself, define the boundaries and present your estimates?
You misunderstood. Wasn’t rebutting, and what you just said confirmed the analogy.
Energy consumption is likely concentrated in the refinery and upstream of it. Not transport of the product to retail outlets.
Obvious example would be tankers carrying 100s of thousands of barrels of gasoline and burning a fraction of that. And that’s downstream of the refinery.
I have been a bear with respect to oil prices for most of the past 10 years. For awhile I doubted my intuition because prices stayed high for so long. Here is a link to a link http://rdcu.be/pfIu which describes many of the reasons. The article does not take into account some important parameters such as interest rates and debt.
Hi Schinzy, last fall, I posted a link to the same or a similar writeup by the same authors here.
I’ve also quoted this before, but it bears repeating:
Of course, money (‘price’) is a symbol created by the human imagination.
From what is understood, after some adjustment, the price of oil (and other commodities) has (have) not fundamentally changed, or changed very little, over decades. On some graphs, it appears like a waveform fluctuating roughly equally above and below roughly zero.
I am not smart enough to understand all of this. But, I guess that since medical costs are going up faster than energy costs we should see that play out first. Right now the government cannot collect enough in taxes to pay for those who cannot afford medical care, and insurers have to raise their premiums so high that many cannot afford the insurance. So, in the near future, most people will abandon seeking medical help [drugs, operations, hospitals, etc] and therefore medical prices will plunge, causing most drug companies, hospitals, doctors, etc. to declare bankruptcy and go out of business. Then we will go back to something like taking a teaspoon of honey and if that does not cure whatever, then you will not be cured because all of the providers will have ceased business. This, of course, will go a long way towards solving the over-population problem.
Bearing in mind Europe spends less for healthcare, and has a higher life expectancy, maybe Americans will live longer from spending less.
Clueless. I don’t understand this stuff either and therefore have pretty much ignored it.
They lost me with the idea that in the not too distant future oil prices will be zero. I can’t wrap my mind around that one.
Hi shallow sand,
That is another glaring flaw in the Hill’s Group analysis. They assume economics is bullshit and that prices are not determined by supply and demand.
As long as a gallon of diesel fuel continues to have about 138,000 BTU of chemical energy, the net energy of that gallon of diesel (which nobody really knows anyway) has no bearing on whether the consumer will be willing to purchase that gallon of fuel. The thermodynamics of all energy used by society is undoubtedly important, but it is not well understood at the societal level.
The price of oil would only fall to zero (or at least become very low like $10/b or less) after high oil prices lead to greater efficiency in the use of oil and substitutes for oil use in transportation are developed. Optimists like Tony Seba think this will happen by 2030, realists like me think more like 2050 (or even 2060 if I am too pessimistic about future oil output), pessimists say such a transition is impossible, but that oil prices will fall due to permanent economic calamity.
Nobody knows.
My guess remains high oil prices from 2020 to 2030 ($100-$150/b), depression from 2030-2037 (lower oil prices due to lack of demand maybe $50/b on average over that 7 year period), then economic recovery and higher oil prices ($125/b in 2017$) from 2038-2050. By that time we might see AVs, and public transportation start to reduce oil demand faster than supply falls and oil prices will start to fall, not that I assume by 2030 that peak fossil fuels will be widely accepted and that governments will spend money appropriately to help transition their economies to non-fossil fuel energy while deficit spending (a la WW2) pulls the World economy out of Great Depression 2.
No doubt this story will be wrong, but that’s how I see it.
Oil and fossil fuels in general will be an energy sink after about 2050 +-10 years. They may still be used for special applications but their usefulness as an energy source will have disappeared.
Probably. There won’t be anymore oil flowing.
Or people.
Agreed! It’s pretty hard to make people flow… 😉
https://en.m.wikipedia.org/wiki/Flow_(psychology)
Named by Mihály Csíkszentmihályi…
… just another run of the mill, ordinary Hungarian immigrant 😉
Personally I believe the whole ETP thing is not even good as a joke, and I haven’t even tried to seriously understand it, since it contradicts just about every verifiable fact I am aware of.
But talking about prices can be tricky, because while two or more people discussing price may be talking about the same commodity, they are all too often either talking about something they know nothing about, or about the price at DIFFERENT stages of the production, distribution, and marketing processes.
I don’t see it happening, ever, as a general thing, but it is at least theoretically possible that the monetary value of oil in the ground, or in a tank above ground, could go to zero. That would be the price the owner of mineral rights could get, or the price the oil field operator could get, ZERO, nothing, no sale.
It probably did happen ,temporarily, from what I read, to some producers of some really low grade crude in places where shipping costs are really high, within the last few years here in the USA. When the so called price, the one every body talks about, the price at a hub, was at the very bottom, it apparently cost more to get this crude delivered than it was worth at the refinery.
Some people, some of them smarter than I am, argue that eventually there will be or might be no market at all for oil.
This is theoretically possible, because renewable energy of one sort or another, or several sorts, might eventually get to be so cheap that it will be possible to manufacture everything currently made from crude from air and water, plus some minor amounts of additives.
I don’t have any difficulty imagining the air and water to oil process being almost totally automated.
But is it at all LIKELY that any of us here will live to see renewable energy too cheap to meter, etc?
In the end, governments often make the final decisions on such issues as what will be done, in general economic terms, in any given country. The market might not say we should give up oil, but it’s possible politicians might.
We can theoretically build out enough renewables to not only give up natural oil, but also to make paints, lubricants,some liquid fuel, and other essential products starting with CO2 and hydrogen from water. Or we might use organic materials as the source of the H and C.
Theoretically we can ,IF we can get “over the hump” and build out enough renewable infrastructure BEFORE fossil fuel depletion or some other problem prevents us from doing so.
But as a practical matter, I would gladly bet ten to one it just won’t happen within the lifetime of any regular member of this forum. . There will be too many OTHER demands on the limited supply of skilled hands and expensive materials in my opinion.
So we will almost for dead sure continue to burn some oil far into the future and use some as an industrial feed stock as well.
Consider the possibility of batteries good enough to run dump trucks all day getting cheap enough to be practical. A BIG farm tractor has an engine with similar horsepower, but it will likely only be needed all day two or three months out of the year. A new one built near the end of the age of crude oil will be serviceable for at least fifty years, assuming parts and fuel are available.
It’s going to be VERY hard to justify scrapping such a machine as a large bulldozer, or fire truck, which needs to run only a few hours from time to time, so long as fuel can be had. It’s hard to imagine it getting cheaper to manufacture fuel from scratch than it is to manufacture it from oil that can be gotten out of the ground at low cost.
And IF the market for oil does collapse, THEN there will very likely be some remaining crude that can be cheaply produced for decades, maybe for a century or longer.
Bottom line, if we get totally away from oil within the next couple or three generations, it will probably be the result of a POLITICAL decision to do so, rather than an economic decision.
Other folks mileage may vary. It could be that I ‘m way to pessimistic, in terms of being worried about industrial civilization and modern life just continuing to exist, never mind believing in a golden age of super cheap super abundant clean energy.
The THERE is there, probably, in technical terms, but there is one HELL of a chasm to be crossed in getting from our present day HERE to that theoretical future THERE.
A successful crossing is not by any means assured.
I did a thermodynamics course in my second year of technical college back in about 1982 and I’ve forgotten just about every detail and equation I ever learned. I guess the idea of teaching electrical engineering students about thermodynamics is that they will have a better understanding of the prime movers that spin the generators that they are concerned with. Having said that, I think I have a pretty good bullshit filter and have never understood the Hills Group’s ramblings about a “thermodynamic basis for the price of oil”. Here’s why.
I am an advocate for pedal to the metal adoption of renewable energy, especially solar (in case anybody doesn’t already know that) which gives me a certain perspective. Let’s say I’m somewhere in the middle east that formerly had more hydrocarbon resources than we knew what to do with but, have basically run out of the good stuff with only difficult to extract stuff left. Let’s say that we have a sizable sovereign wealth fund or other pool of cash from all the years of selling oil. Let’s say that we are in the process of building enough solar to give us more electricity than we will ever know what to do with. Once built, the cost of each additional available unit of electrical energy will be zero or to put it another way, once the solar generating capacity is built there is no point in not using every unit we can.
Let’s say we have enough capacity to power a theoretical electric airplane but, electric airplanes exist only in theory, awaiting the development of a battery with a suitable volumetric and weight energy and power density. wouldn’t we be willing to expend all the excess energy we would otherwise waste, going after some of that difficult to extract gooey stuff if that’s what we need to fly the airplanes we have, that still work just fine on fuels derived from the gooey stuff? See: How many kilowatts to get an electric 747-8 airborne? The people who answered the question at the web page linked to above came up with figures ranging from 90 MW to over 180 MW. Using Jet A1 as fuel, the four turbine engines on a 747 can do the job of producing that power without breaking too much of a sweat! Take a look at the pic of San Diego Gas & Electric’s 30 MW, 4 hour, largest lithium ion battery in the world to get an idea of the dilemma of commercial aviation using batteries. If electric commercial aircraft ever come to pass, they will be very different from the jets we are accustomed to!
IMO, as long as we have excess energy from somewhere and a means of concentrating that energy sufficiently, we will be willing to use it as inefficiently as the surplus will allow, to get whatever it is we have always used energy to do. Whether it be growing vegetables in the winter in Scandinavia using hydroelectric energy or using desalinated seawater to grow tomatoes across 49 acres in South Australia, desalinating water in California or the middle east using solar energy humans will find an economically worthwhile use for any excess energy we have access to. I find it very difficult to believe that, oil will not be worth using some less useful energy source to extract it, in the lifetime of many of us on this web site. Certainly not by 2022 or even 2032 maybe not even 2042.
Additionally I think it is Dennis that keeps making the point that it is the energy in the chemical bonds of the hydrocarbons that we find useful and that it may be possible to extract oil using electric motors rather than combustion engines, in which case the thermodynamics that the Hills Group rambles on about are irrelevant.
I’d like the goal of creating an electric airplane to continue, but we can also solve the problem by making oil-based airplanes so expensive to use that we eliminate all unnecessary use of them. That could happen if fuels get expensive enough. That leaves enough oil, at high prices, to pay for necessary airplane use, and gets rid of a lot of vacation travel, and package delivery which could be handled by railroads and trucks.
Airplane use is popular, but much of it is not necessary to keep economies functioning.
There goes the fresh lobster, shrimp and fish for the middle of the country. There goes the medical emergency flights. The end of doctors without borders. The end of trips to Africa and back – but, that should reduce Ebola concerns. Shuts down skiing in Colorado, honeymoons to Niagra Falls, US weddings in the Carribean. No more professional sports – cannot afford to fly the teams around. Probably ends college sports as well. Even if teams could get to games, visiting fans could not. So much for going to the Cleveland Clinic for a heart problem, or M. D. Andereson in Houston for cancer. Disney goes bankrupt. Cruise lines go bankrupt – people cannot fly to the port cities. Trump remains president until he dies – no one else can afford to fly around to campaign. Cremations sky rocket in popularity – relatives cannot get to a funeral in a timely manner. Serial killers have a field day as more and more people take to hitch-hiking.
Maybe economics will keep on functioning, but these thoughts are arriving faster than I can type them.
Don’t worry, large aircraft are heading toward a double in efficiency and most people forget the power of chemistry to use electric energy to make liquid fuels for use in turbojets.
Dood, what is efficiency for an airplane? Do you know anything at all about this? Do you know the gear ratio for turboprop engines with respect to turbine RPMs?
Do you know what an inertial seperator is? Do you know what semi active seekers are? How about stabilators? What’s the VHF emergency frequency?
Why are you doing this crap?
Not keeping up with the latest research, materials, engine designs and airframe changes are you?
Check out NASA, their latest research claims the ability to cut fuel burn in half for large passenger aircraft.
New concept aircraft like the D8 and blended body aircraft will push efficiency even further.
But I guess you either are just being a twit or don’t believe that long range passenger jets can get to 200 pmpg range.
I do wonder if the extra drag and weight of the turbofan with big gearboxes is really necessary. It’s what we have right now.
I am more interested in smaller, short hop aircraft, which due to lower speed can achieve much higher efficiencies.
You think the future will be like the past? Not even close. One of the best things we could do is make a network of aircraft and airports, short and long haul to replace much of the highway infrastructure. Keep local roads only and fly everywhere else, for passenger transport and light freight hauling. Electric cars and trains do their best in the town and city environments. Long range freight trains would still fit the picture. But flight might just lift us into the next century without wrecking everything.
No, if things hold together, the model of transport and civilization will have to radically change to accommodate a whole different resource and energy system. The old model is a dead end. Time to rethink the whole system and air travel saves a huge amount of energy and material (no roads or bridges in between to build and maintain).
Right now an EV can get 300 mpge at lower speeds. Even the Nissan Leaf can go 300 miles on a charge running slower. So drive to the local airport, air hop to the next state or city, take a car to where you are going.
I’m talking about the marketplace setting the price of airline use. If petroleum rises in price and planes still depend on petroleum, unnecessary uses will disappear as the price goes up.
There will be a in between solution – a hybrid plane:
http://www.airbusgroup.com/int/en/corporate-social-responsibility/airbus-e-fan-the-future-of-electric-aircraft/technology-tutorial/E-Thrust.html
Electric take off and downsized gas power generators for cruise speed for lower fuel consumption.
Perhaps the long run solution will be hydrogen based for greater energy density.
OT for this discussion but, I don’t know where else to post it:
Fishermen save oil survey, says Tullow
Wasn’t this the kind of situation that lead to the discovery of the Cantarell field in Mexico? IIRC the field is named after the fisherman who reported the sheen. Wouldn’t it be ironic if after all the money and high tech wizardry thrown at the search for oil offshore Jamaica, a useful discovery emerges from some good, old fashioned pot luck!
Ugo Bardi just presented a debunking (many contributors) of the Hill’s Group report.
http://cassandralegacy.blogspot.no/2017/02/peak-oil-catastrophism-is-popular-but.html
”Let me just say that it simply makes no sense to start from the textbook definition of entropy to calculate the net energy of crude oil. The approximations made in the report are so large to make the whole treatment useless (to say nothing of the errors it contains). Using the definition of entropy to analyze oil production is like using quantum mechanics to design a plane. It is true that all the electrons in a plane have to obey Schroedinger’s equation, but that’s not the way engineers design planes.”
Would be a nice guest post on POB.
Can we have an Etp exclusive thread so we don’t have to wade through any more of this crap?
Or an Open Thread Petroleum
George actually I agree.
A report that has been thoroughly debunked and characterized as nonsense do not deserve any attention.
Yes, Rune’s and Dennis’ high-energy dirty-model-debunking exergy has gone through the systems and has now produced lots of work and entropy to luxuriate in.
Good show, guys.
[…] clearly shows the many fundamental flaws of the treatment. You can read the results in English by Seppo Korpela, and in Spanish by Carlos De Castro and Antonio […]
Business in the fracking industry seems to be picking up.
http://finance.yahoo.com/news/basic-energy-services-adds-74-110000724.html
So it seems that us bloggers trying to make some sense of peak oil in the political sphere (so as to not wildly mislead readers who are committed to environmental causes, e.g. stopping the Dakota Access pipeline and so on), we need to go back to
“regular peak oil” analysis and jettison the thermodynamic theory material, since it doesn’t seem to be clarifying things for those with an environmentalist commitment, who really do need to understand that the energy crisis still is a “thing.”
Cheers, B.G. in Wisconsin
https://greenpeoplesmedia.wordpress.com/
Hi GreenPeople’s Media.
You said: “we need to go back to
“regular peak oil” analysis and jettison the thermodynamic theory material, since it doesn’t seem to be clarifying things for those with an environmentalist commitment, who really do need to understand that the energy crisis still is a “thing.””
When you say that the energy crisis is still a “thing”, I think you mean a thing that we can, through collective action, somehow overcome. This is not a realistic message to be married to.
You seem to be saying that it would be better not to know the truth if that truth is unpalatable or inconvenient. I do not agree.
I am an environmentalist. I came very close to going to North Dakota myself to put my life on the line to stop the pipeline. But I would have done this knowing full well that even if that important battle could be won, the great human die-off cannot be averted. As environmentalists, we are obligated to protect whatever we can. But we should realize that we are not doing this for ourselves. We will all be gone soon. Even knowing this, I am still an environmentalist.
cunning –
Just out of curiosity. Why would you have waited for years, and after over 100 route modifications, unitl the company completed about 1199 miles out of a 1200 mile pipeline, and after they spent billions of $ to get to that point, to try to stop the last mile? Does that really seem like fairness in America?
If “as an environmentalist” you are “obligated” “to put your life on the line” to “protect whatever” you can, there are houses, roads, water plants, shopping centers, oil wells, gas wells, etc., being constructed every day in this country. I am sure that you can find many that you can go and disrupt. If you disrupt (trespass) on enough, you will eventually run across some deranged contractor who will shoot your ass.
Unfortunately, Mother Nature is clamping down and is going to clamp down in all kinds of ways on all of our deranged asses if and as we keep up our current deathstyle. She will make some of us wish she had a gun or reach for our own to put us out of our misery.
Personal assessment
[translated via Google]
“If something has come to light the appearance of the ETP model, there is a need to equip itself with suitable models that describe the growing non- linearity of the energy system, nonlinearity that will grow if it is not reacted soon to the problems Already detected.
That there is a need to have a model does not mean that any model is adequate, and I think it has or believes that the ETP model is not. However , the ETP model has been received simply uncritically by the community, in a collective error in which I myself have worked in some way. Possibly there has been a confirmation bias : as one colleague of mine, a bright and alert physicist put it, the model started from one of the correct assumptions and came to a conclusion , with which it was reasonable to think that The model worked correctly. The fact is that very few have taken the trouble to analyze the model with the model and to point out its deficiencies. In this sense, it is important to have a critical spirit and not accept without the things that seem to confirm what we believe ; All hypotheses should be examined and all work should be reviewed in order to obtain the best evidence to provide us with the most useful results for all. Possibly, thanks to analyzes similar to this one, I have presented to you that The Hills Group can develop an improved model and with it to improve our tendency the dangerous road that extends before us.
Salu2,
AMT”
Or maybe there’s no need for a model.
Dad: “Sorry, kid… I was going to build a dollhouse for you out of wood and give it a post-and-beam design, complete with different kinds of mini-joints, beams, posts, trusses, joists, dowels and assorted other pieces like tiny windows, etc., that you can assemble, disassemble and reassemble in various ways, sort of like Lego or a Meccano set, but maybe there’s no need for that.”
Kid: “Awww, but Dad!?… Who put you up to this?!”
Dad: “Watcher.”
Kid: “Watcher!? Who’s Watcher?!”
Dad: “Nobody… Or maybe some kind of online model of somebody…”
Kid: “Whaaat?!”
Dad (returning from printer…): “Here, have this nice colorful oil-production-depletion profile-and-forcast graph.”
Nowhere Man (3D group model/instructional music video)
Bah humbug
‘u^
Moving right along, past self proclaimed noble envirowackoism, we have Dead Cow news.
https://www.ft.com/content/031d1674-d74f-11e6-944b-e7eb37a6aa8e
Mr Macri proclaimed a “new era” for the country’s languishing energy sector, which will see foreign companies including Chevron, Dow, BP, Shell and Total, as well as Argentina’s state energy company YPF, invest an initial $5bn in 2017, rising to $15bn in subsequent years.
. . .
In return for the investment commitments from the private sector, the government committed to extending a subsidy being gradually phased out that enables companies to sell gas for three times the international price, with the local price currently fixed at $7.50 per million British thermal units. Mr Macri also announced the elimination of a 15-year-old export duty on oil and oil products.
Ahh supply and demand disciples, another shot to the solar plexus. NatGas.
https://www.bloomberg.com/news/articles/2016-06-03/exxon-ceo-says-argentina-shale-investment-may-exceed-10-billion
The vid in that link is all over reserves replacement failure. Tillerson’s last deal, btw.
I’ve published a new post on projecting the output of existing horizontal US shale wells, available here.
IRAQ PLANS OFFSHORE OIL AND GAS EXPLORATION TO BOOST RESERVES:
http://www.reuters.com/article/iraq-oil-exploration-idUSL5N1GC39E
‘Iraq is planning to start offshore oil and gas exploration to boost the OPEC nation’s reserves, Oil Minister Jabar al-Luaibi said in a statement on Monday.
‘Luaibi said he “gave guidance to the Oil Exploration Company about the importance of exploring territorial waters to assess the hydrocarbons reserves and to boost Iraq’s capacity”.’
Why would a country with a professed R/P of over 90 years, a medium term need to replace declining production and existing issues with balancing the books direct their efforts to finding even more oil? Maybe production would be safer from disruption offshore, or the existing reserves are expensive to develop or they don’t actually have 163 Gb – or all of the above.
Nigeria for some time has had that reputation of offshore safety of flow vs onshore. Iraq headed in that direction, perhaps.
But it is curious, and best we remember that reserves declaration there pointed mostly at surpassing Iran’s claims, not accuracy.
On the Bakken post there was discussion on DUCs. The chart below shows what has happened to the DUCs in September till now. The data is a bit grey because of confidential wells, I’ve assumed any confidential well that has been spudded but shows no ‘runs’ number is a DUC – some may be drilling though, and it means the data for confidential wells is two months old (i.e. depends on production figures rather than the more up to date wells status database). In the 4 to 5 months about 35% have been completed in some way (I don’t know what the one that is drilling means – it might be an error). Mre than I expected have been temporarily abandoned (most of these have passed two years expiry for NC). Almost all the NCW (waivers) are with EOG or CLR – these too are over two years since spud. The average NC age is just over a year, most of the wells over one year are also with EOG or CLR. If these companies have a big campaign of completions, which CLR for sure are planning, then there could be a lot of production coming on line without any increase in rigs.
Yet again I managed to get the wrong image – so try this:
This shows current DUC’s by company with four of the main players highlighted:
[…] Coyne, who published Seppo Korpela’s article here says: “Oil prices are not determined primarily by thermodynamics as the Hill’s Group […]
Art Berman’s latest: The Beginning of the End For The Bakken Shale Play
It’s the beginning of the end for the Bakken Shale play.
The decline in Bakken oil production that started in January 2015 is probably not reversible. New well performance has deteriorated, gas-oil ratios have increased and water cuts are higher. Much of the reservoir energy from gas expansion is depeleted and decline rates should accelerate. More drilling may increase daily output for awhile but won’t resolve the underlying problem of poorer well performance and declining per-well reserves.
Someone told me, a few days ago, that they expected Bakken EUR would decrease by only 4% per year. Can’t remember exactly who that was however. 😉
Well, he has to know better, but phrasing things . . . decline rates should accelerate . . . can’t make sense. The less drilling you do, the slower the per well decline rate, of course. The curves get assymptotic and thus the decline rate decreases.
But what he really meant to say, and almost certainly actually meant, is decline rates for new wells will accelerate. Enno’s famous year by year chart shows this glaringly. The decline slope for 2010, 2011, 2012 wells is far more shallow than the near vertical decline slope of 2014 wells, the last year before company implemented frantic this or that.
Where is that guy who has been hyping technical genius and miracles broadcast in the investor briefings?
If my memory is correct. Some of these Canadian Tar Sand projects need that Bakken oil to dilute. This will eat into production numbers beyond just the Bakken.
Enno Peters’ data for the Bakken does not confirm these declines.
They sort of do if you exclude years past 2014. Look at his chart.
Post 2014 we have speculated on all sorts of choke things and stage count things and whatever out of desperation to maintain loan flow, but 2014 was the last “normal” year and its decline from peak is near vertical on that chart.
Bakken type curves.
source: Oxford Institute of Energy Studies, November 2016
(also using Drillinginfo data)
https://www.oxfordenergy.org/wpcms/wp-content/uploads/2016/11/Unravelling-the-US-Shale-Productivity-Gains-WPM-69.pdf
that ain’t enno’s chart
I could not copy Enno’s chart.
You may see it in his recent Bakken update.
Other sources also show that Bakken wells performance is not deteriorating.
Data for 2015-16 may be distorted by high-grading of the drilling/completion portfolio (activity more focused on sweet spots).
But in any case there was no deterioration in 2015-16.
I could not copy Enno’s chart.
If you have Windows, you can copy anything with Windows Snipping tool. You can even copy from Kindle with Snipping Tool. That’s how I copy Enno’s charts.
But if you have an Apple I would believe Apple has a similar snipping tool.
https://shaleprofile.com/index.php/2017/02/28/projecting-us-shale-oil-production-after-june-2016/
The steepness of the post peak 2014 curve is sharply so vs 2011, and certainly more so than 2010. It’s not even a squint to see it, but 2011 is maybe more legit an examination because of stage count blah blah.
Regardless, newer wells immediate post peak decline is clearly sharper.
{edit here, I think there is a scaling problem on the chart. The raw numbers don’t look like the curve — dunno what that is gonna mean for anything, but the years don’t seem to have the same scale.
By the raw numbers accessible by mousing over Enno’s cool interactive curve the 2014 1 yr decline was 61%, starting and ending December, 2010’s was 54%, 2011 was 57%. Definitely steeper but eyeballs say much steeper than 5%.]
Thank you Ron!
Here are two chart showing Bakken wells quality from 2012 to 2016 from Enno’s shaleprofile (thank you Enno!)
You’re welcome Alex.
Glad to see that you are already an expert user of my site.
Enno,
It’s easy to be an expert user of such an excellent site
Bakken data from Rystad (October 2016):
neither is that
Alex
Thanks for posting the Rystad/Enno charts.
Huge difference in cum curve – especially 2016’s wells – when you view bo versus boe.
coffeeguyzz,
Yes, Rystad data is in boe and reflects rising GOR
How is the EUR calculated? I haven’t seen anything new come out concerning this, and few of the wells have reached exhaustion to be able to give any actual real data. The companies aren’t putting out these numbers and the quoted source, Labyrinth Consulting, is Berman’s company – so it looks like he is calculating the numbers himself without citing the method or assumptions.
EURs are calculated using assumed type curves for the full production cycle of the wells.
Nobody says exactly what are their assumptions.
Therefore, shale companies’ presentations show spectacular improvements in EUR, and Mr. Berman shows spectacular deterioration.
Therefore, shale companies’ presentations show spectacular improvements in EUR, and Mr. Berman shows spectacular deterioration.
And which one do you believe?
none of them.
I think the truth is somewhere in between.
Without significant technological breakthrough (which may not happen), average well productivity and EURs will start to decline, but I do not want to guess when: in 2017, or 5 years from now.
One important thing we don’t know: how many new wells can be drilled in Bakken sweet spots.
One important thing we don’t know: how many new wells can be drilled in Bakken sweet spots.
But there is one thing we do know, they will all be drilled in between laterals that have already been in production for a long time. That is they will all be down spacing wells. Therefore we do know that these wells will have a much lower EUR than the wells to either side of them.
The EUR will not be down by only 4%, not even close. 10% to 20% would be a far more accurate guess.
Ron,
Down spacing affects productivity only when well density reaches some critical point, and new wells start to interfere with existing ones.
The data for 2016 doesn’t confirm that this critical point was already achieved
I had a look at permits for February (admittedly a short month) – 46 new, 16 cancelled – net 30, the same as December and the lowest since around 2006 / 2007, assuming there weren’t many being cancelled back then. Completions are now consistently exceeding spuds, which exceed net permits. So the number of open permits and DUCs are gradually decreasing. There may be some impact from the new pipeline, and a big price jump may do something, but on present trends I’d say permitting will stop some time in the first half of 2018, leaving about 2o00 permits plus DUCs to finish up.
“There may be some impact from the new pipeline”
Dakota Access Pipeline will very likely have crude flowing by April 1, but an early March startup now looks unlikely, a lawyer for the 470,000 b/d project said in court Tuesday.
http://www.platts.com/latest-news/oil/washington/dakota-access-pipeline-very-likely-to-start-oil-27779301
Hi George,
I fit a hyperbolic curve to the data using least squares, when the well profile reaches a 9% annual decline rate (the decline rate gradually decreases on a hyperbolic) I assume exponential decline at 9% per year from that point forward (usually at about 8 years from first output), I assume the well is shut in at an output of 7 b/d, usually this occurs between 20-25 years. Total Bakken/TD EUR for average 2008-2014 well is about 340 kbo over well’s life. For the Eagle Ford the wells have a shorter life and faster decline and EUR is about 240 kb (2010-2014).
For a Bakken 2010-2014 average well Q=12480 b/m, b=1.4, D=0.207 for standard hyperbolic equation:
q(x)=Q/(POWER((1+b*D*x),(1/b))), where q(x) is output in barrels per month and x is month from first output (I use x=0.5, 1.5, 2.5, …).
Hi Ron,
The 4 percent annual rate of decrease in new well EUR was a guess based on several assumptions, that the overall TRR would be about 9 Gb, that the past well performance will be roughly matched for wells drilled through Dec 2016, that about 30,000 total wells will be drilled and that the completion rate is only about 80 wells per month. If the completion rate is lower, say 40 wells per month the EUR decreases more slowly (2%/year), if the well completion rate is higher, say 160 new wells per month then the EUR decrease will be faster (8%/year).
Also if the TRR is lower, the EUR decrease would be higher for any given well completion rate and if the TRR is higher, the the rate of EUR decrease would be lower.
Note that very low URR estimates like that of Verwimp and Jean Laherrere imply very large EUR decreases, like 50% per year. In fact for the URR to be 3 Gb as Laherrere believes, no new wells would need to be completed. There have been 11,000 Bakken wells drilled and 272 kb per well would result in 3 Gb, the average EUR is about 300 kb of oil.
George Kaplan has suggested maybe another 3000 wells or so at most (I believe this is incorrect), that would imply a URR of 4.2 Gb. Proved reserves plus cumulative production is about 7 Gb in the ND Bakken/Three Forks, if we make the unrealistic assumption that probable reserves are zero.
A more realistic estimate is 8 to 9 Gb, which would be 35,000 wells with an average EUR of 250 kb. Or 40,000 wells with an average EUR of 212 kb (average EUR decreases as more wells are drilled because the last 5000 wells would be low quality wells drilled at high oil price levels).
I’m glad to find myself in good company again: ” low URR estimates like that of Verwimp and Jean Laherrere”. It’s about time someone starts a wikipedia page about me too. 😀
Off topic: Dennis, you may choose to explain EUR, URR and TRR from time to time. Newbies on your blog will appreciate that, I believe.
On topic: a decrease of 50% per year seems impossible to you, while documented production history shows an increase of 100% per year for some consecutive years. (An increase of 100% is the same in absolute numbers as a decrease of 50%.)
In the popular press these 100% yoy increases were framed as a ‘miracle’. 50% decreases will probably not be mentioned in the popular press.
Nevertheless, when ‘miracles’ are occuring in a story, nothing seems impossible to me.
Hi Verwimp,
EUR is estimated ultimate recovery and refers to total output from a single well over its life. For the Bakken/Three Forks this has held steady or increased from 2008 to 2013, it is too early to know for sure for more recent wells but the 12 to 36 month average cumulative output has tracked slightly above earlier wells, so far there has been no evidence of a decrease in new well EUR.
TRR is technically recoverable resources from a field such as the Bakken, or from a nation like the US, it is the total resource that could be recovered using current technology if oil prices are high. This is distinct from ERR (economically recoverable resources) which is the portion of TRR that prevailing oil prices result in profitable output. The URR is essentially the same as ERR and stands for ultimately recoverable resources.
Even if no more wells are drilled it is likely that the URR of the Bakken/Three Forks will be more than 3 Gb, just 3000 more wells (4.5 years at the current drilling rate) will bring the URR to 4.2 Gb. Proved reserves plus cumulative production at the end of 2015 would mean a URR of about 7 Gb. Typically for a conventional reservoir probable reserves are about 65% of proved reserves, a very conservative estimate would be zero probable reserves (with a 50% probability of being extracted), the mean would be about 32%. This boosts 2P(proved+probable) reserves to 6.3 Gb, cumulative production is 2.2 Gb at the end of 2015 for an estimated URR of 8.5 Gb if there are no discoveries and no possible reserves get moved to the probable category in the future as oil prices increase. Minimum URR will be 6.3 Gb (90% probability), best estimate (50% probability) is 8.5 Gb with 35,000 to 40,000 total wells drilled (or 24,000 to 29,000 wells left to drill) about 11,000 wells have been completed since 2004 in the Bakken/Three Forks.
Damn Dennis, you have all the bases covered, no matter what happens. If this or this or this happens then we can expect this or this or this. Well hell, how can you possibly miss.
Hey, what the hell is going to happen, that’s what everyone wants to know. Not what will happen if this or this or this happens.
Okay, so you don’t know. Hell, I don’t either.
Take care.
Ron
Hi Ron,
I have never claimed clairvoyance. I create a model and show what might happen depending upon assumptions. The point was if only 80 wells per month are completed, the EUR will decrease at about 4% per year in a scenario with a URR of 8 Gb. The EUR is guessed to start decreasing in June 2017 and gradually increase to the maximum rate over a 12 month period reaching the maximum rate in June 2018. If the well completion rate changes the EUR increases (with an increase in completion rate) and or decreases (with a decrease in the completion rate).
I get different URRs in my model for any given total number of wells (15,000, 25,000, or 35,000) by changing the rate of decrease in new well EUR.
Essentially the well profile shifts lower while maintaining the same overall shape .(Q_naught decreases in the hyperbolic equation).
If the well completion rate changes the EUR increases (with an increase in completion rate) and or decreases (with a decrease in the completion rate).
Dennis, I do believe we are talking about estimated EUR per well. Are we not?
So lets just wait just a cotton picking minute here. If more wells are drilled, meaning they must resort to drilling in more marginal areas, then the Estimated Ultimate Recovery increases? No, hell no! If more marginal wells are drilled then the EUR must decrease. If, on the other hand, they drill fewer wells, meaning they drill only the very best areas, then the EUR would have to increase. No? If not, then why not?
What is wrong with my reasoning here Dennis? Please tell me.
Hi Ron,
I wrote that incorrectly as is clear from the example I had given earlier:
If the completion rate is lower, say 40 wells per month the EUR decreases more slowly (2%/year), if the well completion rate is higher, say 160 new wells per month then the EUR decrease will be faster (8%/year).
In the comment above I incorrectly said what you quoted, but should have said:
If the well completion rate changes the annual rate of decrease of EUR increases (with an increase in completion rate) and or decreases (with a decrease in the completion rate).
I mistakenly left out the part in italics above.
A faster completion rate will increase the rate of decrease of new well EUR so that EUR will fall faster and a decrease in the completion rate would have the opposite effect.
Also you said:
If, on the other hand, they drill fewer wells, meaning they drill only the very best areas, then the EUR would have to increase.
This is not what happens in my view, if fewer wells are drilled, EUR will not increase, I assume once the EUR starts to decrease it will continue to do so as long as new wells continue to be completed (unless there is some technological breakthrough which I do not expect).
Lower completion rate, lower rate of EUR decrease
higher completion rate, higher rate of EUR decrease
Dennis – That is not what I said. I said 3000 more in the core. There may be many more in other areas outside, but there would be a step change down in EUR per acre (for clarity: not per well because they would probably have to be really long laterals to be economical), and hence cost per barrel. The EUR in this area is not really known because the E&Ps were still drilling wildcats here when the USGS did their analysis.
Hi George,
In David Hughes analysis there he claimed that it was pretty clear at that point where the core area was, you should reread his analysis, it is pretty good.
I know you said 3000 more in the core, but you seem to imply that very few wells will be drilled outside the core.
I doubt there will be a step change, I imagine they will gradually move from the core and work outwards (from the “center” of the area) and the EUR will gradually decrease as they move to more marginal areas. In addition it will not happen all at once over time there will be a gradual move from core to outside the core so the average EUR of wells that start producing in say 2018 would see a gradual decline because the mix of core and non-core wells will gradually change.
I am taking the average of all wells, it seems unlikely that one day all wells will be completed in the core and the next day all wells that are completed will be outside the core.
If that happened, we indeed would see a step change in the EUR of the average well. 🙂
RIG FIRM SEADRILL SAYS MAY FILE FOR CHAPTER 11 BANKRUPTCY:
http://uk.reuters.com/article/seadrill-results-idUKL5N1GD1KX
“Q4 EBITDA beats forecast, Q1 EBITDA seen down …Rig firm Seadrill, battling with $14 billion in debt and liabilities, said on Tuesday it may have to file for Chapter 11 bankruptcy protection if it fails to reach a restructuring agreement with its lenders.”
How can they pay back $14 billion when all the deep sea drillers are cutting prices just to stay alive? I think the offshore drillers are probably the canary in the coalmine for the problems in the oil industry. With the E&Ps pulling back from high cost / high risk offshore plays (possibly just because of price, but maybe because they are running out of prospects), a lot of the older projects and in-fall options having been used up in the high price years from 2010 and discovery rates falling their future is not great. Even if prices pick up it is going to take time for the E&Ps to start designating capital for high risk plays and then will their be the equipment and expertise available? An even if there is will they actually start finding enough big plays to go keep going. I think there are fewer than 220 deep see oil projects at the moment, maybe a bit fewer for gas. About 70 oil are under construction, another 30 are simple tie backs, the rest are expensive projects that were around in 2013, and didn’t go ahead. Shell seems to be keen to proceed on some – Vito, Kaikias, Appomatox – Statoil has a few in the works, but they will run out of larger projects soon; BP and Total are definitely going for gas rather than oil. Overall though I think the big drilling companies are going to have to start whittling down, maybe merging, a few, and that process might continue and till they all disappear.
The whole offshore rig industry is really in the deep; especially the companies targeting the deepwater segment like Seadrill. Too many rigs were coming into the market based on borrowed money. Now, the activity level is more or less approaching 40% utilization rates with dayrates more than halved compared to 2014. There is too little scrapping being done to balance the market near term, and I cant blame the companies for wanting to cold stack their dear assets as opposed to scrapping. The market will return someday, but for the moment this is a no go industry for casual investments in stocks -unless you really know what you are doing.
As regards the mechanism of doom, which of course is oil scarcity, there is an accelerant — population growth. ZH had an addendum article this week on that:
http://www.zerohedge.com/news/2017-02-27/demographic-panic-china-considering-birth-rewards-encourage-citizens-have-more-babie
“Due to one of the lowest birth rates in the world (1.5p per family), thanks to the one child policy, China is now considering offering incentives to its citizens to get out there and ‘screw for China’, a la Denmark.
Source: Reuters
The potential move was revealed by Wang Peian, vice-minister of the National Health and Family Planning Commission at a social welfare conference on Saturday, the newspaper said on Tuesday.
Birth rates rose to 17.86 million in 2016, the highest level since 2000, after the country issued new guidelines in late 2015 allowing all parents to have two children amid growing concerns over the costs of supporting an aging population.
”
Chart says by 2050 India, China and the US combined will add about 500 million people to world population, and even this won’t be the primary source of growth — Europe, Russia and Japan will have modest declines overwhelmed by the growth in Africa (Nigeria 138%!!!) and South America.
Should have fairly sharp pop declines way before then, but who would ever say that from the UN?
Maybe they should encourage immigration as a way to build population.
That’s not exactly popular in most countries worth moving to these days. Several not very nice folks have profited from the discontent (Trump. LePen, Wiilders, etc.)
But if China wants more people, accepting immigrants is one way to do it and would set China apart from those countries who are closing the doors to them and trying to throw them out.
Who likes immigrants. Even the US don’t really like them besides the big towns, afro-americans and mexicans don’t have THAT good reputation – and islamic people, especially with low education aren’t liked that much, too.
I think letting immigrate several million africans, mexicans and muslims into the USA, without any selection, just first come first serve, would bring even the USA to the brink of civil unrest in a few years.
Yep, people like you didn’t like the Germans, the Irish, the Polish, the Japanese, the Chinese, the Indians. Yet they are all here and have been here for a long time now.
Get over it.
When you like them so much, why do they live if ghettos in the USA most times and not in normal neighborhoods?
The last time I visited the country I got warnings to visit their quarters – not really a sign they are welcome.
And why have I seen them mostly in low paying jobs when they are so good integrated?
There IS a problem, and you can’t just go over it with a ” I need more people, just tell everyone to come”. Look at the history of multi-cultural nations about history – most have been hold together with military, and have broken up to parts after the central government failed to never reunite again. Stable nations have been build around ethnicies in history – perhaps today are other times.
The chinese empire broke down several times in history, was conquered multiple times, was split in warlord kingdoms – but always reunited.
You don’t have this with austria – after the Habsburg Monarchy resigned, only the core was left. The conquered rest just split off to never come back – the people had nothing in common with the core country.
And when today immigration doesn’t work really good for the USA, THE immigration nation, how good will it work for China, a nation that has no experience in immigration, to welcome a 100 million of low education immigrants – you won’t get other – to beef up population.
The United States was built by immigrants. They have come in waves. It often takes several generations for them to move up into the middle class, but they do.
Each new wave starts out living in the cheaper parts of town, but then they make money, integrate more fully, and a new group of immigrants moves into those low cost neighborhoods until they rise up.
Now we see that much of Silicon Valley’s success is do to immigrants. If the US becomes too unwelcoming, these same people may choose to stay in their own countries, or go elsewhere. Canada is hoping that more immigrant entrepreneurs choose to locate there.
EIA has GoM for December up almost 50 kbpd to 1728 (2.8% on the month, 7.7% y-o-y). Not quite the record set in 2009, but close.
http://www.eia.gov/dnav/pet/hist/LeafHandler.ashx?n=PET&s=MCRFP3FM1&f=M
But total U.S. production was down 91 kb/d in December, following a cumulative increase of 337 kb/d in October-November.
As we know from NDIC data, North Dakota output was down 89 kb/d
Oil production in Texas declined by 17 kb/d. Texas data for November was revised down by 23 kb/d.
Year-on-year changes (Dec16 vs. Dec15):
Total U.S.: – 442 kb/d (-4.8%);
North Dakota: -201 kb/d (-17.6%);
Texas: -193 kb/d (-5.8%)
GoM: +123 kb/d (+7.7%)
Revised monthly data vs. projections in the February STEO
Thanks George and Alex. I do not know why so much non-oil related BS gets on this site.
Worth noting EIA may get defunded soon.
We will have to concentrate on IEA, Jodi and probably best, BP.
With the December production data available now, we can see that 2016 total daily GOM production was at a record high – about 1.6 mmbopd, vr. the previous highs of about 1.56 bopd in 2002 and 2009 (and 1.55 in 2010).
It is not just the new projects contributing to this, but new production coming from some of the bigger, older fields – Mars/Ursa, Atlantis, Mad Dog and Tahiti have all seen increases in production in recent years.
2017 will be a slow year for brand new big projects (though I am sure a number of tiebacks will come on line), but I believe these legacy assets, along with new projects from the last few years that George Kaplan has been tracking, will keep production levels on a par with 2016 levels.
Vague recall is hurricanes in the past could take 75K bpd or 100K bpd off of GOP production averaged over a full year.
And we haven’t had signif hurricanes there for a long time. That hurricane lack effect has added production.
That sounds about right. The last major significant hurricane impact year was 2008 (Gustav and Ike) – September production that year was below 300 kbopd. 2005 (Katrina-Rita) also had measurable hurricane related downtime.
In their predictions, the EIA averages that out such that every year has about the same amount of minor hurricane related downtime.
I wonder if there is an industry participant that knows a lot about US oil inventories that visits this site. After 2 years, I am beginning to wonder whether much of increased oil inventory in the US is actually required in order to have the system as it now exists to function properly.
Speculators get in and out of positions, usually rather quickly when “sentiment” changes. Therefore, I would have expected several times during the past 2 years to see plunges in US inventory levels. That is, speculators dump their physical positions to refiners, who in turn cut off imports for say a month. But, imports seem to keep rising. Oil imports are up 5% this year, while at the same time gasoline and diesel invetories are above the 5 year range. And, even a recent article called for the crude oil price to plunge based upon sluggish gasoline demand. Something just does not feel right to me. It is like speculators keep adding to long physical positions for 2 years.
Suppose HFT engines take over the oil market.
Watcher says: “Suppose HFT engines take over the oil market.”
Does “supposing” use any energy at all?? What is the EROEI of a suppose?
… Depends on the thermodynamics.
It’s non trivial. The plastic in the H F T keys required energy to manufacture. Then diesel for the trucks to transport the keyboard.
It’s a disaster, I tell ya.
On the topic of Bill Hill, here is a link to a column on Alice’s blog that emphasizes some of his comments made on the PO.com forum.
http://energyskeptic.com/2017/shale-light-tight-oil-from-the-etp-model-qa/
One of his comments is that it takes 74,000 BTU to get one gallon of gas to the consumer. I guess that might mean to the gas station.
Bill certainly has collapse-appeal, and plenty of folks seem to like that.
I’ve never seen decent EREOI type computations. They all have their assumptions and the assumptions never seem to become an industry standard.
So people talk about 1.5:1 and 8.3:1 and it can’t mean anything at all.
Better maybe to note qualitatively the effort to haul a horse drawn wagon of lumber out to a site, drill a hole, and get 15,000 bpd from it 100 yrs ago.
Versus now you build a 40 story tall gizmo with a gazillion horsepower marine engine on it, motor 6000 miles to a site, drill a hole, and get 15,000 bpd from it.
Whatever the EREOI number is, it ain’t trending in the right direction.
Nobody expects EROEI to increase for extractive industries.
Why not? Isn’t that what automation does?
And btw sportsfans, factoid in the era of automation:
Avg productivity growth in the US 1995-1999 — 1.84%. (dates chosen to capture PCs in the workplace, but avoid dotcom pop)
Avg productivity 2012 – 2016 — 0.49%. That’s with smartphones, Uber, telecommuting, ZIRP and less proportional labor costs given the decline in participation rate. And oil scarcity.
Watcher says: “I’ve never seen decent EREOI type computations.”
What is the most significant difference between EREOI and EROEI?
Sorry, I am now bored with this thread.
I frequently mistype that. Or not.
So maybe the model doesn’t fit the real world. For all practical purposes oil is a finite resource. The thermodynamic part was just the efficiency that isn’t get any better and is calculated roughly. The net energy left over to run our world has nothing to do with thermodymics. Total energy= energy to get oil + waste energy in using oil + energy left over to use. So I’m my opinion this guy is getting way lost in the insignificant minutiae of waste energy.
Agree with this assessment. I have done free energy models before and the Hills Group analysis is clearly misguided in getting lost in irrelevant detail.
The FAO food index for February came out today – up 0.9 or 0.5%.
http://www.fao.org/worldfoodsituation/foodpricesindex/en/
There is a very strong correlation between the index and oil price, as below. The fit is best when the prices are concurrent by month (i.e. with no delay). Correlation with a deflated index, using 2% per year, but non-deflated WTI price is even closer (charts further below). Effectively the food price is directly related to oil price, but also grows independently of oil from year to year.
I think the food index at around 200 is when social unrest starts to accelerate. Given the close correlation shown this may be as much to do with energy prices as food prices (but local subsidies may play a role in which is most important). For 2017, index of 200 would occur with oil at $71. The price would then have to decline at over 3% per year to prevent the food index continuing to increase. Given I think there will be a supply crash sometime in 2018/ 2019 I think falling prices will only happen if there is major disruption in the world economy. So it might come down to food in developing countries versus jobs and wealth in the OECD. I don’t think a switch to natural gas (which is usually closely correlated to oil in price anyway; sometimes directly within the supply contracts) or renewables can happen fast enough to make a difference in that time frame.
This shows correlation with deflated food index at 2% (which is slightly lower than the FAO uses but gives a closer fit with oil price alone, this may be to do with using a non-deflated oil price but I’m not certain). Note also that the scales are not adjusted exactly to match the correlation (although close) but give a tighter fit in recent months.
Oil and food prices has a high correlation, going back at least too the 1970. However, food price spikes have occurred when oil prices and low yield (e.g. droughts in food exporting nations) occur in the same year. Global harvests have been high for several years and stocks are at all-time high. It will probably take 1 or 2 years with below average harvests to trigger a new price spike.
If I remember correctly, the current upswing is mainly a result of problems with sugar export from Brazil. Grain prices are still low.
An oldie but goldie on food prices and political instability: Lagi, 2011. The Food Crises and Political Instability in North Africa and the Middle East. http://necsi.edu/research/social/food_crises.pdf
If I remember correctly, the current upswing is mainly a result of problems with sugar export from Brazil. Grain prices are still low.
Which is really due to long term drought in South Eastern Brazil where most sugarcane plantations are located.
220 is when things got smashy the last couple times around.
http://static4.businessinsider.com/image/50b22be86bb3f77832000009-960/food-prices-riots.jpg
YO MIKE…EXXON LOVES THE CHILIE
Exxon Mobil shifts investments to quick-earning shale
http://www.worldoil.com/news/2017/3/1/exxon-mobil-shifts-investments-to-quick-earning-shale
I never get tired of being right, do you ever get tired of being wrong?
Tee tee, you shouldn’t have skipped school this morning; there is no e in chili.
Exxon has always been a bunch of no oil finding geniuses (Guyana was its first big one in a long time) and have run out of ideas around the world. W. Texas is perfect for them; it does not have to look for anything. It paid 6.6 billion dollars for some goat pasture, (get somebody to help you research BOPCO on Enno Peters sight) but might be able to squeak out 1.25: 1 ROI’s out of that stuff because they won’t have to borrow money to do it.
While you and whomever are on Enno’s sight, research N. Mexico HZ Permian, Bone Spring, Cline, etc., and you’ll see that of 3,180 wells drilled since 2009, only six have UR over 400K BO. That’s a about what it takes to reach payout at 48 dollar WTI.
Don’t forget to check that mailbox for checks today!
That may sound good to investors, but if Exxon actually does greatly increase short-term oil production from US shale, it will suppress oil prices. It is a no-win for the oil industry. And if “U.S. shale will absorb 50% of Exxon’s worldwide drilling budget” then the company has run out of other places to drill.
Essentially, then, when the Permian runs out, Exxon will likely be in major decline.
Most IOCs and large independents now seem to be acknowledging that they are running out of oil prospects and that their overall yearly oil production isn’t going to be growing much, if at all . Despite rising oil price the international oil rig count has stayed low, even still falling slightly. Total, ENI and BP are moving to gas. BP have said their production mix will move from 1:1 oil:gas to 1:1.5 by the mid 20’s. Marathon and CoP are retreating (or have already retreated) internationally. They, with Chevron and ExxonMobil are concentrating on US LTO. Shell are finishing off what outstanding discoveries they have, but it’s hard to see where they go after that. Canadian oil sands is really slow with nothing much being approved. Only Anadarko and Hess seem to be still in the old pattern of exploration and development (and their debt load is pretty big). Iran and Iraq was a big hope but has been pretty quiet for the IOCs recently, (there was something about Majnoon expansion, and increased offshore exploration). Maybe there’ll be some announcements from Iran soon on redevelopment plans, which won’t, however, involve US companies by the look of things.
Do you understand two things?
1) this is not an investment blog. It’s a peak oil blog, because, gasp, that’s the title of the website.
2) Exxon failed to organically (via exploration, not acquisition) replace reserves produced last year, and the year before, and the year before
Do you understand these two things? It’s a yes or no question.
From the http://www.worldoil article there is a quote that by 2025 XOM’s US shale holdings could reach 750,000 bpd. I assume that means BOPD??
I would like to see someone estimate how many wells that will take. I guess they need to add 25,000 to 30,000 to reach 750K BOPD in 2025, assuming most the wells will be in the Permian Basin.
So, using 25,000 wells at $8 million per well, including all the necessary facilities, I come up with US shale CAPEX of $200,000,000,000 from 2017-2025 to reach that figure, plus a minimum of another 2,000 wells per year to maintain that figure. Or $16 billion per year in CAPEX thereafter.
That, of course, assumes no service price inflation, which is highly unlikely if XOM is that aggressive, as I still do believe in supply and demand, contrary to what Ron may think (LOL I am kidding Ron).
XOM’s current market cap is $337 billion. That is the whole company, some 4 million BOEPD plus refining plus chemicals plus pipelines plus everything else.
Why would they want to spend over half of the market capitalization of the company to achieve 750K BOPD of output when the market does not value the WHOLE company I have generally described above in a similar multiple.
Ah yes, I forgot, the shale miracle/revolution, which has provided consumers low gasoline prices and has caused US shale E & P’s to have record earnings per share. Oh wait, did I get something wrong in the preceding sentence?
If anyone wants to challenge my back of envelope calculations as to how many wells XOM needs to put online from now to 2025 to be producing 750K BOPD from US shale in 2025, have at it. I am still waiting on a challenge to my 2015-2017 oil and gas price back of napkin payout calculations. Do I hear crickets chirping?
I made a post but it got lost in cyberspace.
The XOM article says that XOM may have 750K bpd in US shale production by 2025. Assuming that means BOPD and not BOEPD, I estimate XOM needs to spend $200 billion in US shale CAPEX to reach 750K BOPD by 2025 and needs to spend at least another $16 billion annually thereafter in said CAPEX to maintain said production rate. This assumes no price inflation.
XOM’s market cap is $337 billion. This includes 4 million BOEPD of production, plus refining, plus chemicals, plus everything else.
So, is $200 billion for 750K BOPD of US shale a good investment decision for XOM between now and 2025.
I suppose it depends on the price of oil, which apparently does not matter to TT and US shale.
I love the quote that US shale is arguing with itself. It needs higher prices to be profitable, but is working like hell to put on more supply, which reduces prices. LOL!!
As always, Shallow, you do a much better job than I do of putting things in financial perspective. If one looks at the wells that the Bass Brothers were drilling on this big block that Exxon bought, they were weenie wells, at best. Why Exxon expects to do better is beyond me. It reminds me of Apache and its Alpine High “Discovery.” Everyone from Tom Brown to Claytie to EOG tried that, and bailed. Now, given its first 9 wells on this block, Apache will have to bail too. 2G BO reserves my Texas ass.
Some people think its “un-American” to be brutally honest about our hydrocarbon resources. In fact its un-American not to be.
I am astonished the shale oil industry does not hate one another with a passion; it is competing with each other for reduced, and declining CAPEX, the more production EOG puts on the market, the more it hurts Continental. Rinse and repeat. Today a 750 mile pipeline was proposed from the Permian to Corpus Christi, which will devastate Eagle Ford oil prices. This shale oil thing is now way out of control. Again. If I were not IN the oil business it would indeed be humorous, all of it. I may nevertheless write a book about it someday. I have a title in mind but it is not quotable here.
Mike. I assume the block assembled by the Bass family has many tracts that date back to Syd Richardson in the 1930s.
So, this family, who is already loaded, and has likely held on to a lot of this acreage for decades, suddenly decides to sell during a down time in the oil market?
The Bass Family knows how much this acreage is going to cost to develop, I just estimated that above. I agree they ended up with a load of XOM stock, so they don’t think XOM is dead. However, they are now liquid and rather than owning acreage requiring loads of CAPEX, they now own a boatload of one of the largest and most liquid companies in the world for that matter. They can time their sales of bits and pieces.
The Bass family, as I recall, took Uncle Syd’s oil fortune and multiplied it several times over. They are sharp and have some sharp folks working for them. Not a reputation of giving stuff away.
I am sure XOM sees prices doubling some day, and would assume they will develop this acreage in a more sane way than some of their junior peers. Helps it is mostly HBP.
SS with regard to EXXON deal you maybe missing a key component.
Think back, what were the stated reasons Exxon bought XTO.
Clue: was it for the reserves or was it for the in-house expertise or both.
The reality is if you listen to the players, is that the learning curve as well as the expertise within any given basin can take millions of $$$ to gain and then to build on and apply to any given formation. My guess is the Bass’s came to the same conclusion exxon did a decade ago, they do not have the expertise or the people to developed their minerals (Economically) and while they had the money they did NOT want to build a LTO company from the ground up. In the oil patch scale and expertise are going to continue seperate the haves from the have nots.
With regard to price mattering, this march 1st looks a hell of a lot better than last, I am a glass half full kind of guy, you have got be…. to be successful in this business. I bet that the Saudis have plugged back in their calculator. Market share means NOTHING if you are going broke. They will keep prices up by limiting production just like they always have. It’s a win win they sell less of their the oil for more$$$. IN another year or so the market will be under supplied or prices will rise. Either way I can buy chili with my LTO checks ?
But if US production happens as fast as the oil companies claim it will, then it doesn’t matter what the Saudis do. The US alone will keep prices down.
As I keep saying, all this planned drilling is going to result in real or perceived excess supplies. Drilling as much as US companies want and as fast as they want will keep down prices.
On the other hand, if we see each of the LTO areas peak, THEN prices will rise, but it won’t be good.
shallow sand,
Below is a chart from Exxon’s presentations.
It’s kboepd.
How realistic is this? Seems very optimistic to me.
How many wells.
BOEPD I agree will need a lot less wells than I stated using 6/1 mcf to BOE conversion.
Talking about cluelessness. The geniuses at the WUWT blog (where Javier and other deniers get their talking points from) claim that they have discovered the logistics function should replace the Hubbert curve. After much discussion, some commenter finally pointed out that the Hubbert curve IS the logistics function. Not only that, but it looks like they used the cumulative version of the logistics (i.e. the sigmoid or S-function version), not realizing the implications of this.
https://www.bloomberg.com/news/articles/2017-03-02/saudi-arabia-still-bears-brunt-of-oil-cuts-as-opec-output-drops
SAUDI ARABIA STILL BEARS BRUNT OF OIL CUTS AS OPEC OUTPUT DROPS
“Riyadh pumps 90,000 barrels a day less in February
Previously compliant Angola ramps up output at new fields”
Is Saudi cut entirely voluntary? Venezuela’s almost certainly isn’t. Angola has at least another 200 kbpd ramp up on two new projects to come, so I doubt they’ll take any more notice of the cuts.
“Iraq’s production dropped by 50,000 barrels to 4.44 million barrels a day, the survey showed.”
“Iran’s output increased to 3.83 million barrels a day, slightly above its goal of 3.797 million barrels a day. … Libya and Nigeria — both exempt from the accord — saw combined 50,000 barrel-a-day growth.”
Iraq might have peaked, if only as a pause. Libya and Nigeria growth has slowed, maybe near the limit.
I’m very interested to see what kind of an annual peak in domestic consumption they experience this year. As well as if they respond with increased production. The next couple years are going to be interesting times in KSA.
Since there’s no new “Open Thread: Petroleum,” I’ll just leave this super-optimistic story from CNBC here.
Lately it seems as if the business news about U.S. oil and gas is written with this blog P.O.B. in mind, as a refutation of what y’all are saying here.
If this story premise were true (and I don’t think it is) it would be a refutation of both “conventional peak oil” and the “thermodynamic model(s)” because it seems to imply that even with a sh*tty EROEI as the shales have, it takes a Really Big Corporation to make that EROEI pay off, but it can be done !
http://www.cnbc.com/2017/03/03/oil-majors-move-deeper-into-shale-country-bumping-up-us-production.html?__source=yahoo%7Cfinance%7Cheadline%7Cheadline%7Cstory&par=yahoo&doc=104316095&yptr=yahoo
I found the below on oilpro.com about eog. I am sl8ghtly confused since a 8% increase in wells is forecasted to cost at least 42% more; even considering ducs this seems strange and does not bode well for their 2018 results. Any insights?
Capex in 2017 should range from US$3.7 to US$4.1 billion, with 480 net wells targeted for completion this year, from 445 wells in 2016. Capex in 2016 was U S$2.6 billion.
Well costs should range from “flat to lower” versus last year, with EOG striking a positive note on continued efficiencies and service contract expirations, offsetting potential cost increases.
New sherif in town:
EPA withdraws information request for the oil and gas industry
http://www.worldoil.com/news/2017/3/3/epa-withdraws-information-request-for-the-oil-and-gas-industry
???
Cunning linguist, why don’t you hop on over to Peakoil.com, we can use someone well versed in the technical aspects of ETP, to interject some incontrovertible facts into our discussions. You must get bored of naysayers here discounting Etp with hardly any sound reasoning or grasp of its methodology. Regards
Hi Cm.
Thanks.
I don’t really get bored of the naysayers here because I am not technically here. I am officially banned for life from peakoilbarrel.com. Every comment I make could be my last. The only reason I am here now is because peakoilbarrel is part of a well organized hatchet job against the Etp model. I dropped in with my new IP blocker to complain and they allowed my comments to be visible because they need someone to argue with or their scheme would look even sillier than it does. They are just trying to use me (hopefully I have frustrated their effort somewhat). I am sure that they will reinstate my ban soon enough and I will disappear from this forum without any notice. Then they can get back to business: producing an endless stream of oil industry propaganda.
I would really love to be a part of the ongoing conversation at peakoil.com, but I can’t. I used to comment there as Futilitist, but I was banned. Then I commented as SumYunGai, but I was once again banned. Since then, I tried to open another account, but they are now using cookies to detect my IP address, so I have been unable to make any comments and they banned that user name also.
Peakoil.com has the same agenda that this place does. It is very obvious.
It is Onlooker. Yes I do remember you. You didn’t take no bull from no one. Well, I can say that PO.COM has become boring, a few of us plus Short conversing with moronic trolls. Thanks for the heads up about this site. I am afraid the Oil Industry has many hired hands and accomplices , so this surprises me not. Our whole progression in these limits to growth environment has been painfully pathetic. Our species scarcely wishes to acknowledge these grave problems much less tackle them. Sad
Hi Onlooker.
You said: “Our species scarcely wishes to acknowledge these grave problems much less tackle them.”
I would say that our species scarcely wishes to acknowledge these grave problems because there is no way to tackle them. Think about it.
The site hits for peakoil.com have been plummeting ever since I was banned. Does correlation equal causation in this case? Hmm…ask AdamB.
Please tell everyone left on the peakoil.com Etp Q&A thread that Futilitist/SumYunGai says hello and I wish I could be there (sort of). But since I can’t, please feel free to post any of my comments from here to there. It will drive Tanada nuts!
“This ETP report such as it is is not a true model that has a run time to predict the future it is just a couple of equations wrongly applied that give a set of false results. ” I replied to this as follows ——-This is just wrong. First, the Etp model from what I gather is not meant to be predictive. Nevertheless, the data it produces allows one to reach assertive conclusions. One can do that by accepting certain self evident premises
First: Modern economies depend on abundant energy to function
Second: FF especially oil are the prime sources of energy for Modern economies
Third: The Oil Industry must provide sufficient net energy to the General Economy to justify continued operations exemplified by the notion that when the whole process of providing energy to the the Economy expends more energy than it provides, the exercise becomes a futile one
So, the Etp attempts to account for all costs including the thermodynamic/entropy ones and then looks at the production history to determine when the oil industry will cease to be a viable industry and by extension when society can expect to encounter serious/devastating economic turbulence. Nothing fraudulent or mystifying about all this
Oh and apparently the Etp has been quite accurate as an oil price predictor
” If the authors have a better handle on how much energy is expended in oil production, they can form the EROEI ratio and it would constitute an independent check on the work of Hall and his coworkers on EROEI. Such an independent analysis would be valuable.”
It would indeed be valuable, since the work of Dr Charles Hall is unreliable. Hall’s work contains repeated, severe, mathematical errors which invalidate his analysis. I just wrote a blog post about this.
http://bountifulenergy.blogspot.com/2017/03/dr-charles-hall-is-still-totally-wrong.html
-Tom S